Correct option is C
The Mössbauer effect is the emission and resonant absorption of nuclear gamma-rays studied under conditions such that the nuclei have negligible recoil velocities when gamma-rays are emitted or absorbed. This is only achieved by working with solid samples in which the nuclei are held rigidly in a crystal lattice. The energy, and thus the frequency of the gamma-radiation involved, corresponds to the transition between the ground state and the short-lived excited state of the nuclide concerned.
We illustrate the study of the Mössbauer effect by reference to 57Fe spectroscopy. The basic apparatus includes a radioactive source, a solid absorber with the 57Fe-containing sample and a gamma-ray detector. For 57Fe samples, the radioactive source is 57Co and is incorporated into stainless steel; the 57Co source decays by capture of an extra-nuclear electron to give the excited state of 57Fe which emits gamma-radiation as it decays to its ground state. If 57Fe is present in the same form in both source and absorber, resonant absorption occurs and no radiation is transmitted. However, if the 57Fe in the source and absorber is present in two different forms, absorption does not occur and gamma-radiation reaches the detector. Moving the source at different velocities towards or away from the 57Fe absorber has the effect of varying the energy of the gamma-radiation (i.e. by the Doppler effect). The velocity of movement required to bring about maximum absorption relative to stainless steel (defined as an arbitrary zero for iron) is called the isomer shift of 57Fe in the sample, with units of mms-1.
The isomer shift gives a measure of the electron density on the 57Fe centre, and isomer shift values can be used to determine the oxidation state of the Fe atom.
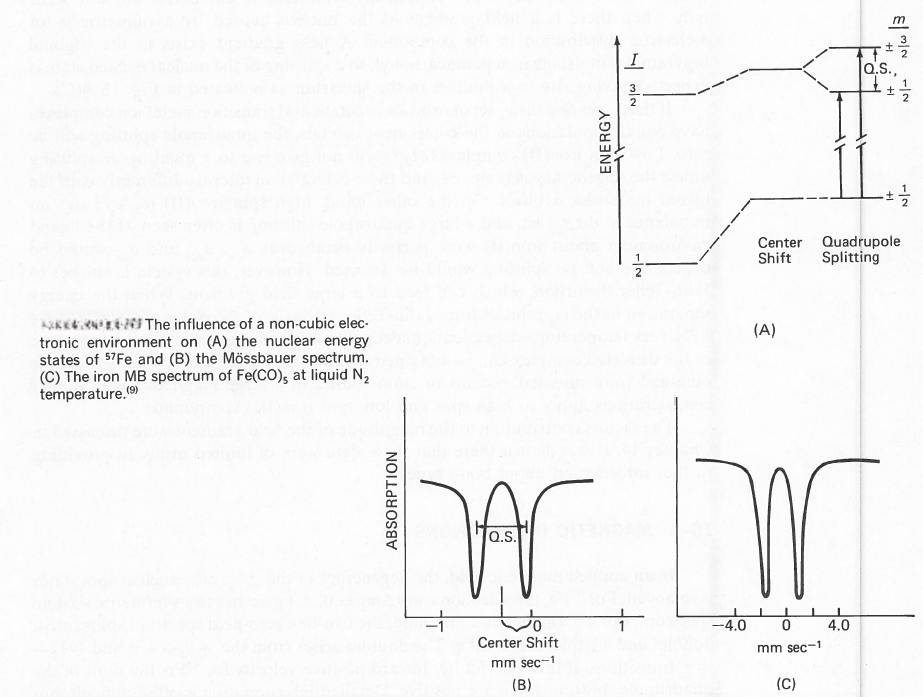
For non-integral spins, the splitting does not remove the + or - degeneracy of the mI levels, but we obtain a different level for each ±mI set. Thus, the electric field gradient can lead to I + ½ different levels for half-integer values of I (e.g., two for I = 3/2 corresponding to ±½ and ±3/2). For integer values of I we obtain 2I + 1 levels (e.g., five for I = 2 corresponding to 2, 1, 0, - 1, -2). The influence of this splitting on the nuclear energy levels and the spectral appearance is illustrated in figure for 57Fe. The ground state is not split but the excited state is split, leading to two peaks in the spectrum. The center shift is determined from the center of the two resulting peaks. When both the ground and excited states have large values for I, complex Mössbauer spectra result.