Correct option is B
The Isolobal Analogy
The isolobal analogy was developed by Roald Hoffmann in 1982. According to Hoffmann two fragments are isolobal if the number, symmetry properties, approximate energy and shape of their frontier molecular orbitals and the number of electrons in them are similar not identical but similar. Some of the reactions of the metal carbonyls are parallel with main group non-metals and compounds. For example, chlorine atom and methyl (CH3.) free radical both have 7 valence electrons, one short of a noble gas configuration. Three of sp3 orbitals of carbon are involved in the formation of sigma-bonds with the hydrogens. The fourth sp3 orbital is singly occupied and has higher energy than the bonding ones. The Mn(CO)5 fragment has 17 valence electrons, one short of the 18-electron. The sigma-bonding between Mn and CO ligands in this fragment may be considered to involve the five of d2sp3 hybrid orbitals of Mn. The sixth hybrid orbital is singly occupied and has higher energy than the five sigma-bonding orbitals. Hoffmann points out that any fragment with a singly occupied orbital of a sigma-type is a free radical and may be able to form structures similar to those found for methyl radical. Halogen atom and methyl radical exhibit chemical similarities with 17-electron organometallic fragments like Mn(CO)5.
The isolobal analogy is expressed by a double headed twirly arrow
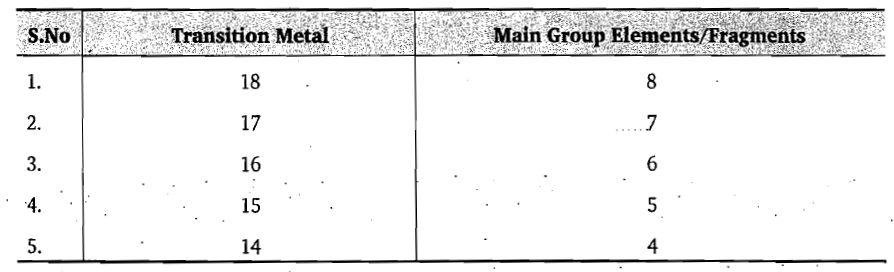
A. [BH]: Boron is a three electron donor. H is a one electron donor. Total electron count=4
B. Fe(CO)3: Fe is a eight electron donor. CO is a two electron donor. Total electron count=8+6=14
C. [P]: Phosphorus is a five electron donor.
D. Co(CO)3: Co is a nine electron donor. CO is a two electron donor. Total electron count=9+6=15







