Correct option is C
Electron Counting in Carbonyl Clusters
Many carbonyl clusters have structures and are electron deficient similar to higher boranes BnHn2-. Therefore, the approach used to describe the bonding, structure and electron count for higher boranes can be used for carbonyl clusters and other clusters. Wade gave the method for electron counting in HNCC. The structure of the cluster is decided purely by the number of cluster electrons (called skeletal electrons), not by any other factor. It is a general feature of closo- BnHn2- anions that there are no B-H-B or BH2 group and each B-H bond is a normal 2e bond requiring one electron from B and one electron from H. As boron starts with three electrons, it has two electrons left to contribute to the cluster and these electrons are regarded as skeletal or framework electrons. Thus, BnHn2- anions have 2n + 2 skeletal electrons (the 2n electrons from nB atoms and the 2 electrons from the anionic charge). In borane clusters, each BH unit has an sp orbital that points directly towards the centre of the cluster and the other two px and py orbitals that point along the surface of the cluster. The nB atoms have 4n atomic orbitals which are always distributed as follows: n in the n(B-Ht) bonding orbitals, (n + 1) in cluster bonding molecular orbitals, (2n - 1) in non-bonding and antibonding cluster molecular orbitals. In BnHn cluster, there are (n + 1) cluster bonding molecular orbitals and 2n electrons contributed by n B-H units, two additional electrons are required for BnHn2- ion. This is the reason why the BnHn2ions are more stable than BnHn itself. Since the shape of the cluster depends only on the number of skeletal electrons, it is possible (conceptually) to remove a vertex group, BHt from the cluster leaving two skeletal electrons that the vertex BHt group was contributing. This is possible only if BH2+ is removed instead of a BHt unit leaving one vertex of the cluster empty. If BH2+ unit is removed from ion is obtained. This will have the same polyhedral structure because the electron count has not changed but one vertex is now unoccupied. The neutral B5H9 borane can be obtained by adding the appropriate number of protons (four protons in this case), this does not alter the number of electrons in the system because protons (H+) are the zero electron species. Hence all the bonding molecular orbitals are just filled. The protons bridge the faces of polyhedron which include the missing vertex. In general, the number of skeletal electron pairs in .
The boranes are classified as: closo, nido, arachno, hypho and klado boranes. The boranes in which each corner is occupied by B atoms are called closo boranes. These have the formula BnHn2-.The number of vertices and skeletal electron pairs are respectively n and (n + 1). This indicates that the vertices in a closo borane will be one less than the number of skeletal electron pairs. If one B atom is removed from a vertex of a closo structure, a nido (Latin: nest like) structure will result. The nido boranes have the formula
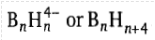
. The number of vertices will be (n - 1) of an n cornered polyhedron and the number of skeletal electron pairs is (n + 2).
If two vertex B atoms are removed, an arachno (Greek: spider's web like) borane will result. With two vertices missing, the structure is even more open than in the nido structure. Arachno boranes have the formula
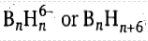
. The number of vertices will be (n - 2) of an n cornered polyhedron and the number of skeletal electron pairs is (n + 3).
The hypho (Greek: net like) boranes have the formula
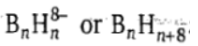
.They have (n - 3) vertices leaving three vertices unoccupied of an n cornered polyhedron and the number of skeletal electron pairs is (n + 4) .
The klado (Greek: branched) boranes have the formula
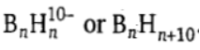
.They have (n - 4) vertices leaving four vertices unoccupied of an n cornered polyhedron. The number of skeletal electron pairs is (n + 5).
The method for calculation of skeletal electron pairs is called the Wade's rule.
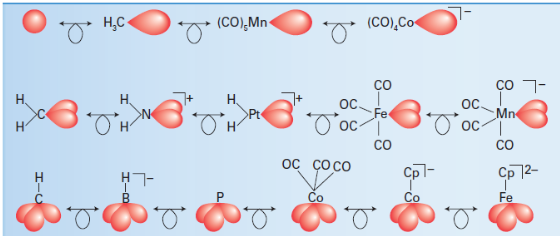
In current terminology, the structurally analogous fragments are said to be isolobal, and the relationship is expressed by the symbol
.The origin of the name is the lobe-like shape of a hybrid orbital in a molecular fragment. Two fragments are isolobal if their highest energy orbitals have the same symmetry (such as the σ symmetry of the H1s and a Csp3 hybrid orbital), similar energies, and the same electron occupation (one in each case in H1s and Csp3).Table lists some selected isolobal fragments and the first line shows isolobal fragments with a single frontier orbital. The recognition of this family permits us to anticipate by analogy with H−H that molecules such as H3C−CH3and (OC)5Mn−CH3can be formed. The second line of Table lists some isolobal fragments with two frontier orbitals, and the third line lists some with three.
B4H10=[B4H4]6-=arachno
[B8H8]2-=closo
[B10H14]=[B10H10]4-=nido
[Mn(CO)4B3H8]:Mn(CO)4 (15 electrons) can be written as BH2 (five electrons). Thus, [BH2B3H8]=[B4H10]=[B4H4]6-=arachno







