Correct option is A
Vaska's complex is the trivial name for the chemical compound trans-carbonylchlorobis(triphenylphosphine)iridium(I), which has the formula IrCl(CO)[P(C6H5)3]2. This square planar diamagnetic organometallic complex consists of a central iridium atom bound to two mutually trans triphenylphosphine ligands, carbon monoxide and a chloride ion.
Vaska's complex can undergo oxidative addition and is notable for its ability to bind to O2 reversibly. It is a bright yellow crystalline solid.
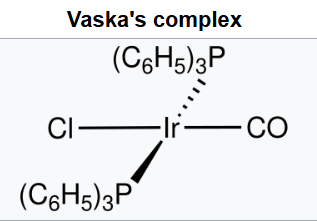
Studies on Vaska's complex helped provide the conceptual framework for homogeneous catalysis. Vaska's complex, with 16 valence electrons, is considered "coordinatively unsaturated" and can thus bind to one two-electron or two one-electron ligands to become electronically saturated with 18 valence electrons. The addition of two one-electron ligands is called oxidative addition. Upon oxidative addition, the oxidation state of the iridium increases from Ir(I) to Ir(III). The four-coordinated square planar arrangement in the starting complex converts to an octahedral, six-coordinate product. Vaska's complex undergoes oxidative addition with conventional oxidants such as halogens, strong acids such as HCl, and other molecules known to react as electrophiles, such as iodomethane (CH3I).
Vaska's complex binds O2 reversibly:
The dioxygen ligand is bonded to Ir by both oxygen atoms, called side-on bonding. The resulting dioxygen adduct reverts to the parent complex upon heating or purging the solution with an inert gas, signaled by a colour change from orange back to yellow.







