Correct option is A
Reactions between metal carbonyls and cyclopentadiene usually yield mixed ligand complexes, e.g. Fe(CO)5 reacts with C5H6 to give (η5-Cp)2Fe2(CO)4. Two isomers of (η5-Cp)2Fe2(CO)4 exist , cis and trans, and both have been confirmed in the solid state. The Fe-Fe bond length (253 pm) is consistent with a single bond giving each Fe centre 18 electrons.
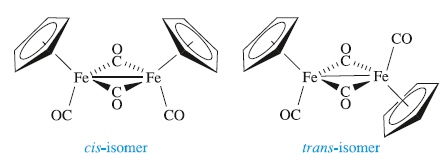
In solution at 298 K, both the cis- and trans-forms are present and the terminal and bridging ligands exchange by an intramolecular process.
The protons of the two cyclopentadienyl ligands in both isomers (cis- and trans-) are chemically equivalent in solution. Both the cis- and trans-isomers lead to identical proton environments for the η5-C5H5 protons due to the fast exchange between the two isomers, making the proton environments indistinguishable.







