Correct option is C
Dissociative Substitution
General mechanism of dissociative reactions is given below:
The first step is the slow dissociation of a CO ligand. This step is rate determining and independent of the concentration of incoming ligand Y. The rate law for this reaction:
Rate=K1[Complex]
In some cases, the back reaction, K-1, becomes important and the intermediate LnM partitions between the forward and backward reactions. The rate of reaction is affected by increasing the concentration of ligand Y because K2 now competes with K-1. The rate of reaction (1) is :
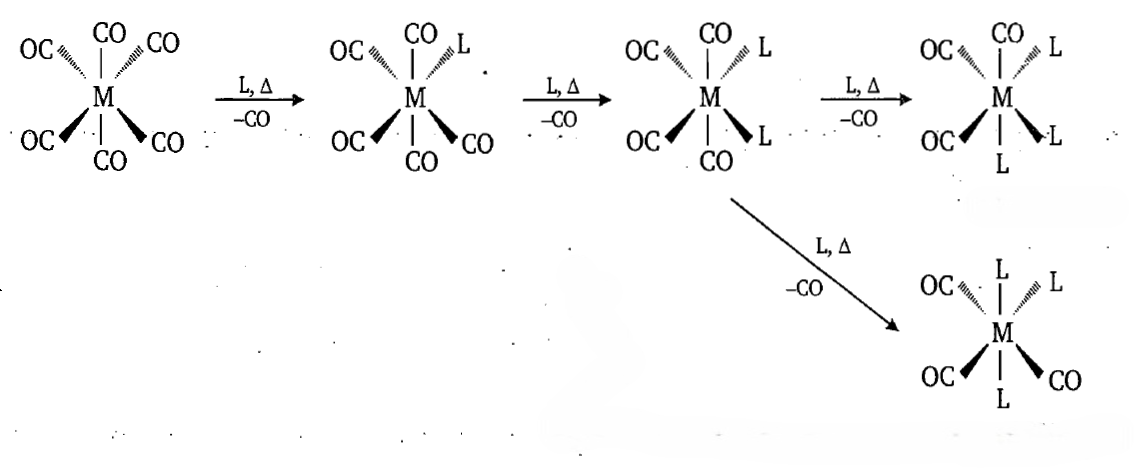
Subsequent substitutions by the incoming ligands like phosphines, PR3 always occur cis to the initial ligand. This is because the substitution of any ligand that is a better sigma donor but a poorer pi acceptor than CO will lead to an increase in the back bonding between metal and the CO ligand trans to the incoming ligand. The substitution reaction rarely proceeds further than fac- M(CO)3L3 because the electron density on the metal would be too great.
The facial isomer in which the incoming PR3 ligands occupy a triangular face of the octahedron is preferred to the meridional isomer in which the ligands occupy a meridian. This is because COs have a higher trans effect than that of phosphines. Therefore, substitution continues until there are no COs trans to a CO.
Oxidative addition inserts metal atoms into single bonds
Potential ligands that do not have a lone pair or filled pi type orbital are still able to interact with transition metal complexes but only by breaking a sigma bond. This is the first step in a wide variety of processes and is described as oxidative addition because the formal oxidation state of the transition metal is raised by two, for example, M(0) to M(II), in the process. This is the result of having two extra ligands bearing a formal negative charge.
The number of coordinated ligands also increases by two so the starting complex is usually in low oxidation state and coordinatively unsaturated, that is, it has an empty site for a ligand, whereas the product is usually coordinatively saturated, that is, it cannot accept another ligand unless it loses one first.
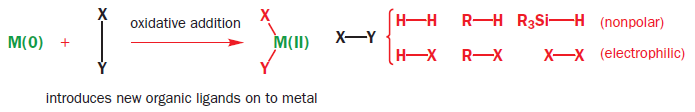
In product A, bromine is added in a cis fashion, consistent with concerted formation of the two new osmium-bromine bonds. The Os(0) complex becomes a new Os(II) species.







