Correct option is C
A cyclopentadienyl complex is a coordination complex of a metal and cyclopentadienyl groups (C5H5-, abbreviated as Cp-). Cyclopentadienyl ligands almost invariably bind to metals as a pentahapto (η5-) bonding mode. The metal–cyclopentadienyl interaction is typically drawn as a single line from the metal center to the center of the Cp ring.
Biscyclopentadienyl complexes are called metallocenes. A famous example of this type of complex is ferrocene (FeCp2), which has many analogues for other metals, such as chromocene (CrCp2), cobaltocene (CoCp2), and nickelocene (NiCp2). When the Cp rings are mutually parallel the compound is known as a sandwich complex.
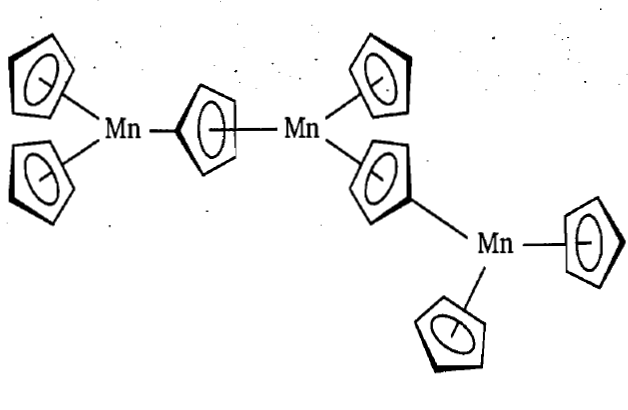
Manganocene, Cp2Mn unlike the other metallocenes, is dimorphic. It is polymeric in solid state and consists of infinite chains of CpMn fragments bridged by cyclopentadienyl rings as shown in figure. At high temperature it is isomorphous with ferrocene. It is ionic, high spin d5 and formulated as Mn2+2Cp-.