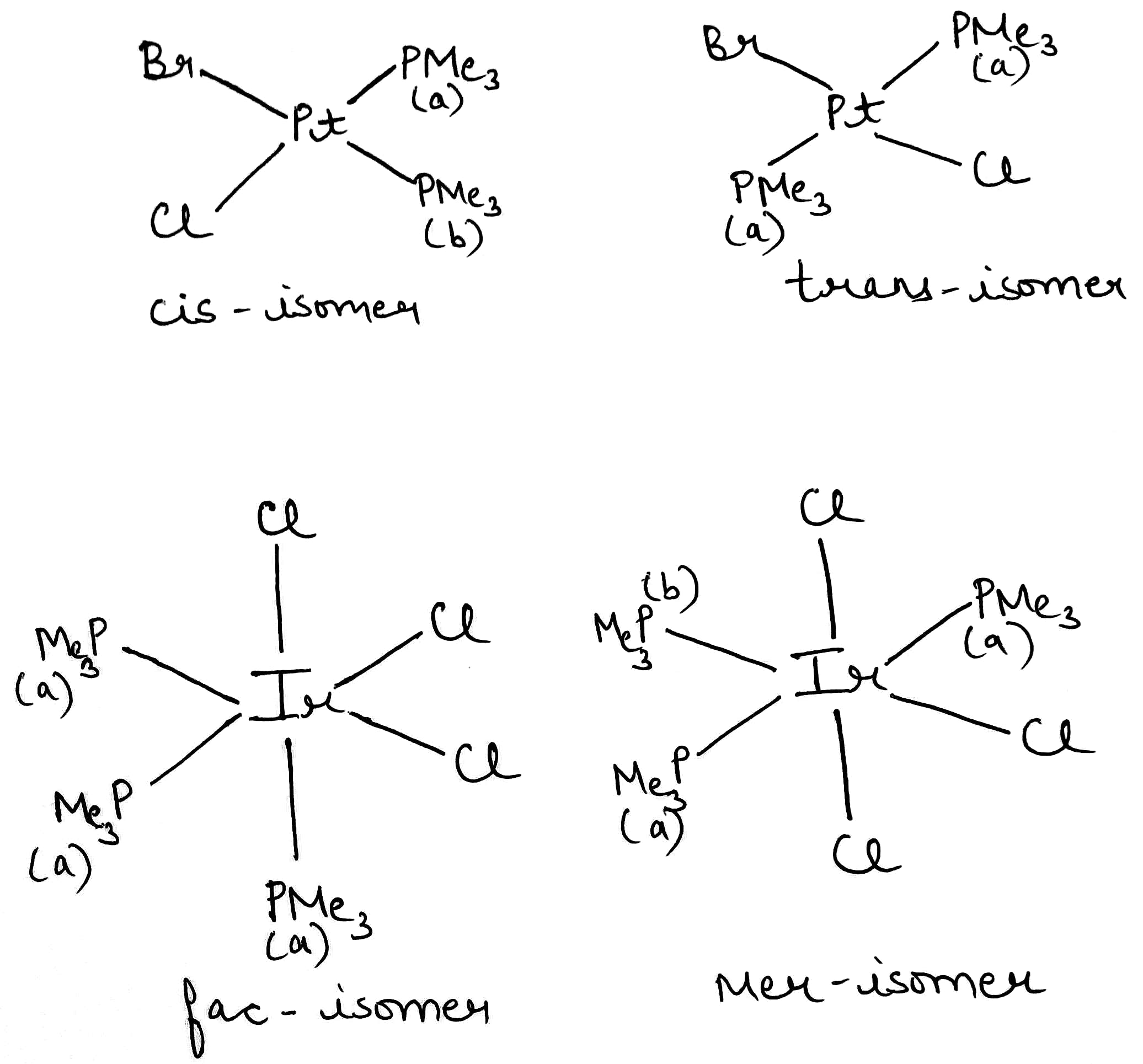
Correct option is B
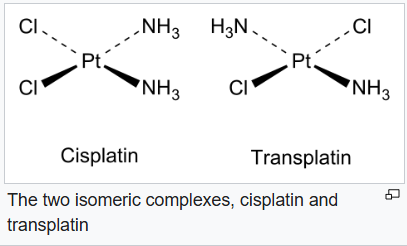
Cis–trans isomerism, also known as geometric isomerism, describes certain arrangements of atoms within molecules. The prefixes "cis" and "trans" are from Latin: "this side of" and "the other side of", respectively. In the context of chemistry, cis indicates that the functional groups (substituents) are on the same side of some plane, while trans conveys that they are on opposing (transverse) sides. Cis–trans isomers are stereoisomers, that is, pairs of molecules which have the same formula but whose functional groups are in different orientations in three-dimensional space. Cis and trans isomers occur both in organic molecules and in inorganic coordination complexes.
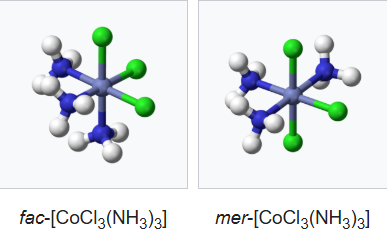
A related type of isomerism in octahedral MX3Y3 complexes is facial–meridional (or fac–mer) isomerism, in which different numbers of ligands are cis or trans to each other. When three identical ligands occupy one face of an octahedron, the isomer is said to be facial, or fac. In a fac isomer, any two identical ligands are adjacent or cis to each other. If these three ligands and the metal ion are in one plane, the isomer is said to be meridional, or mer. A mer isomer can be considered as a combination of a trans and a cis, since it contains both trans and cis pairs of identical ligands.
Phosphorus-31 NMR spectroscopy is an analytical chemistry technique that uses nuclear magnetic resonance (NMR) to study chemical compounds that contain phosphorus. Phosphorus is commonly found in organic compounds and coordination complexes (as phosphines). Solution 31P-NMR is one of the more routine NMR techniques because 31P has an isotopic abundance of 100% and a relatively high gyromagnetic ratio. The 31P nucleus also has a spin of 1/2, making spectra relatively easy to interpret.
The cis isomer of complex A will give two signals due to different chemical environment of the two PMe3 groups. The trans isomer of complex A will give one signal.
The fac isomer of complex B will give one signal. The mer isomer of complex B will give two signals due to different chemical environment of the two PMe3 groups.