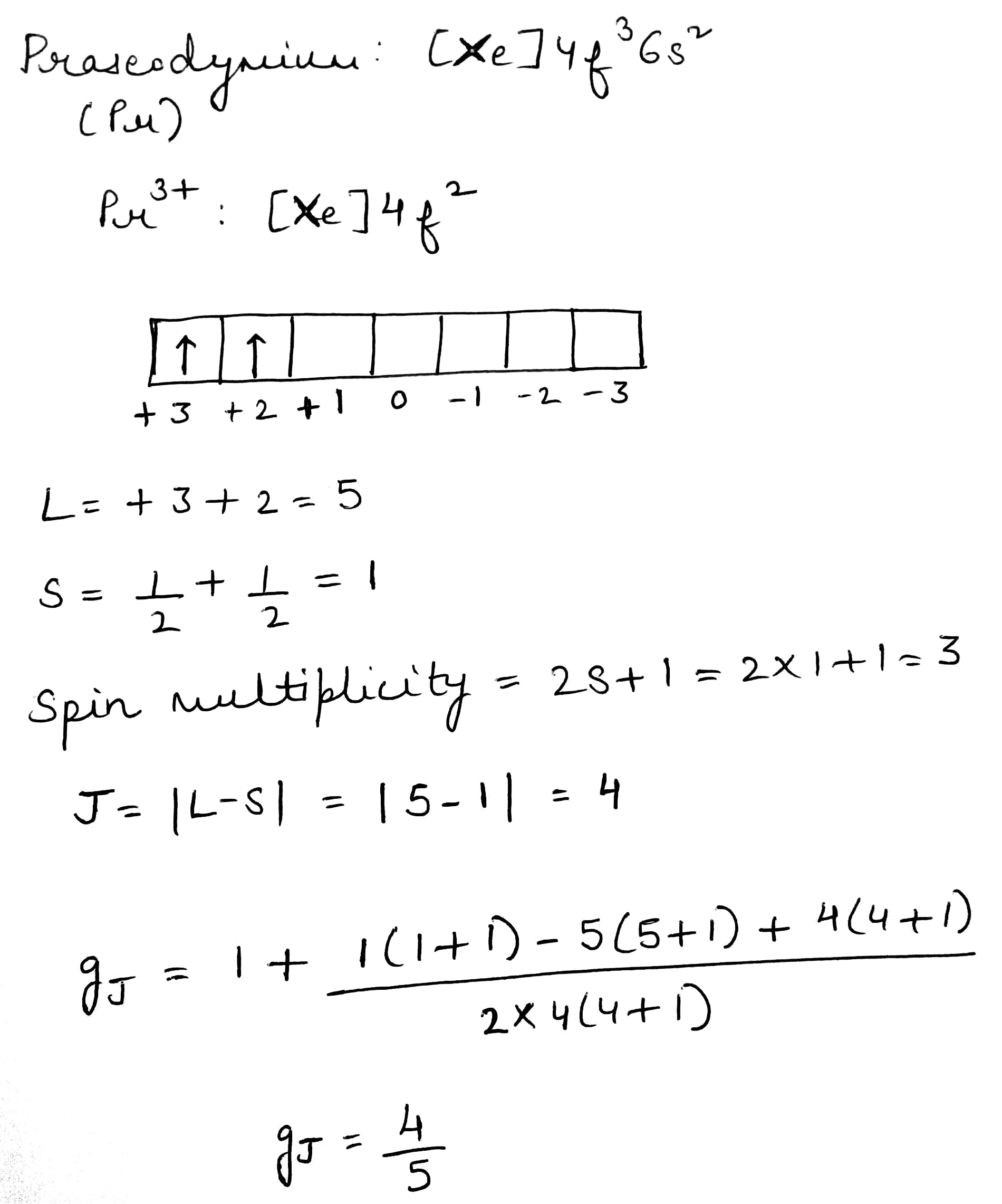Correct option is D
The notation for a full term symbol is:
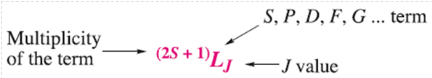
For any system containing more than one electron, the energy of an electron with principal quantum number n depends on the value of l, and this also determines the orbital angular momentum which is given by equation
The energy and the orbital angular momentum of a multielectron species are determined by a new quantum number, L, which is related to the values of l for the individual electrons. Since the orbital angular momentum has magnitude and (2l+1) spatial orientations with respect to the z axis (i.e. the number of values of ml), vectorial summation of individual l values is necessary. The value of ml for any electron denotes the component of its orbital angular momentum, ml(h/2π), along the z axis. Summation of ml values for individual electrons in a multi-electron system therefore gives the resultant orbital magnetic quantum number ML:
Just as ml may have the (2l+1) values l, (l-1) ... 0 ...-(l-1), -l, so ML can have (2L+1) values L, (L-1) ... 0 ...-(L-1), -L. The allowed values of L can be determined from l for the individual electrons in the multi-electron system. For two electrons with values of l1and l2:
The modulus sign around the last term indicates that allowed values of L are 4, 3, 2, 1 or 0. For systems with three or more electrons, the electron–electron coupling must be considered in sequential steps: couple l1and l2as above to give a resultant L, and then couple L with l3, and so on. Energy states for which L=0, 1, 2, 3, 4... are known as S,P, D, F, G... terms, respectively. These are analogous to the s, p, d, f, g... labels used to denote atomic orbitals with l=0, 1, 2, 3, 4... in the 1-electron case. The resultant orbital angular momentum for a multi-electron system is given asmay only be zero or a positive value. As an example, consider a p2configuration. Each electron has l=1, and so the allowed values of L are 2, 1 or 0. Similarly, for a d2configuration, each electron has l=2, and so the
The spin quantum number, s, determines the magnitude of the spin angular momentum of an electron and has a value of 1/2. For a 1-electron species, ms is the magnetic spin angular momentum and has a value of +1/2 or -1/2. We now need to define the quantum numbers S and MS for multielectron species. The spin angular momentum for a multielectron species is given by equation, where S is the total spin quantum number.
The quantum number MS is obtained by algebraic summation of the ms values for individual electrons:
For a system with n electrons, each having s=1/2, possible values of S fall into two series depending on the total number of electrons: S=1/2,3/2,5/2....(for an odd number of electrons) and S=0,1,2...(for an even number of electrons)
S cannot take negative values. The case of S=1/2 clearly corresponds to a 1-electron system, for which values of ms are +1/2 or -1/2, and values of MS are also +1/2 or -1/2. For each value of S, there are (2S+1) values of MS: Allowed values of MS: S, (S-1), ... -(S-1), -S. Thus, for S=0,MS=0, for S=1,MS=1, 0 or -1, and for S=3/2, MS=3/2, 1/2, -1/2 or -3/2.
The interaction between the total angular orbital momentum, L, and the total spin angular momentum, S is defined by the total angular momentum quantum number, J.
The following equation gives the relationship for the total angular momentum for a multi-electron species.
The quantum number J takes values (L+S), (L+S-1)....L-S, and these values fall into the series 0,1,2...or 1/2,3/2,5/2. The method of obtaining J from L and S is based on LS coupling that is spin-orbit coupling.
The value of J for the ground state is given by (L-S) for a sub-shell that is less than half-filled, and by (L+S) for a sub-shell that is more than half-filled.
The magnetic moment μ of many d-metal ions can be calculated by using the spin-only approximation because the strong ligand field quenches the orbital contribution.
In a free atom or ion, both the orbital and the spin angular momenta give rise to a magnetic moment and contribute to the paramagnetism. When the atom or ion is part of a complex, any orbital angular momentum is normally quenched, or suppressed, as a result of the interactions of the electrons with their nonspherical environment. However, if any electrons are unpaired the net electron spin angular momentum survives and gives rise to spin-only paramagnetism, which is characteristic of many d-metal complexes.
For orbital angular momentum to contribute, and hence for the paramagnetism to differ significantly from the spin-only value, there must be one or more unfilled or half-filled orbitals similar in energy to the orbitals occupied by the unpaired spins and of the appropriate symmetry (one that is related to the occupied orbital by rotation round the direction of the applied field). If that is so, the applied magnetic field can force the electrons to circulate around the metal ion by using the low-lying orbitals and hence it generates orbital angular momentum and a corresponding orbital contribution to the total magnetic moment.
For the lanthanoids, where the spin-orbital coupling is strong, the orbital angular momentum contributes to the magnetic moment, and the ions behave like almost free atoms. Therefore, the magnetic moment must be expressed in terms of the total angular momentum quantum number J:
Lande g factor and μ is the Bohr magneton