Correct option is D
The metal dxy, dyz and dxz atomic orbitals (the t2g set) are non-bonding in an [ML6]n+,σ-bonded complex and these orbitals may overlap with ligand orbitals of the correct symmetry to give п-interactions. Although п-bonding between metal and ligand d orbitals is sometimes considered for interactions between metals and phosphane ligands (e.g. PR3 or PF3), it is more realistic to consider the roles of ligand σ*-orbitals as the acceptor orbitals. Two types of ligand must be differentiated:п-donor and п-acceptor ligands.
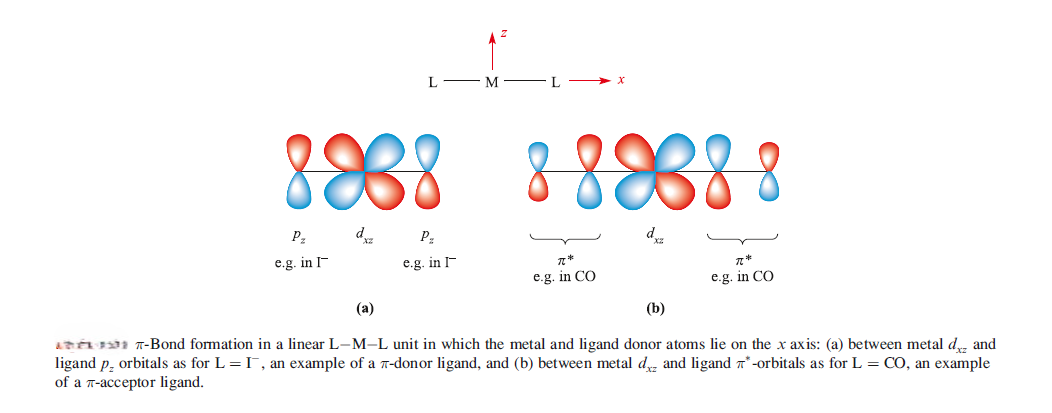

Example of pi-acceptor ligand is CO and the metal–ligand pi-bond arise from the back-donation of electrons from the metal centre to vacant antibonding orbitals on the ligand. Pi-Acceptor ligands can stabilize low oxidation state metal complexes.
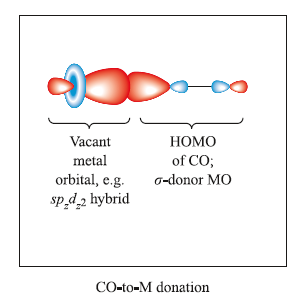
The figure below shows the sigma-interaction between the highest occupied molecular orbital of CO (which has predominantly C character) and a vacant orbital on the metal centre (e.g. an spzdz2 hybrid).As a result of this interaction, electronic charge is donated from the CO ligand to the metal.
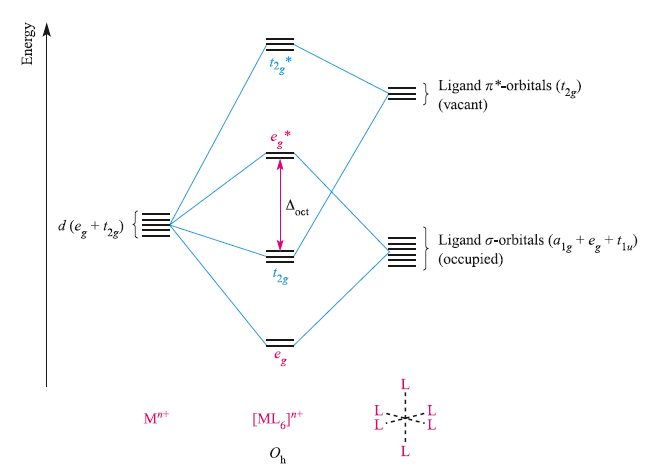
The figure below shows the pi-interaction that leads to back-donation of charge from metal to ligand.
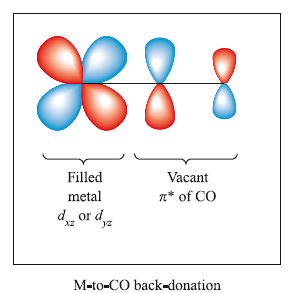
This ‘donation/back-donation’ bonding picture is the Dewar–Chatt–Duncanson model. Carbon monoxide is a weak sigma-donor and a strong pi-acceptor (or pi-acid) and population of the CO π*-MO weakens and lengthens the C-O bond while also enhancing M-C bonding.
Resonance structures for the MCO unit also indicate a lowering of the C-O bond order as compared with free CO.


In the IR spectrum of free CO, an absorption at 2143 cm-1 is assigned to the C-O stretching mode. The lower the value of
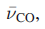
, the weaker the C-O bond and this indicates greater back-donation of charge from metal to CO.
The presence of positive charge on the metal carbonyl complex causes
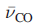
to be higher than in free CO. In such complexes, the C-O bond distance is shorter than in free CO and the M-C bonds are relatively long.
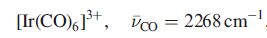
For A,
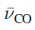
is 2101cm-1.







