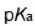Correct option is D
Meldrum's acid or 2,2-dimethyl-1,3-dioxane-4,6-dione is an organic compound with formula C6H8O4. Its molecule has a heterocyclic core with four carbon and two oxygen atoms.
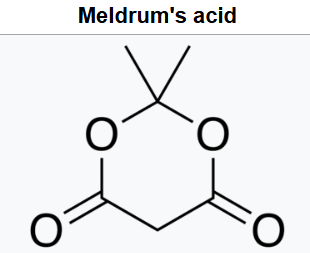
The compound can easily lose a hydrogen ion from the methylene (CH2) in the ring (carbon 5); which creates a double bond between it and one of the adjacent carbons (number 4 or 6), and a negative charge in the corresponding oxygen. The resulting anion [C6H7O4]- is stabilized by resonance between the two alternatives, so that the double bond is delocalized and each oxygen in the carbonyls has a formal charge of −1/2.
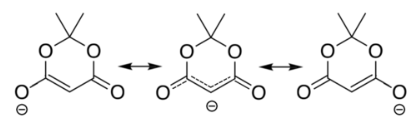
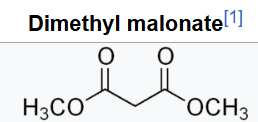
The ionization constant pKa is 4.97; which makes it behave as a monobasic acid even though it contains no carboxylic acid groups. The unusually high acidity of this compound was long considered anomalous—it is 8 orders of magnitude more acidic than the closely related compound dimethyl malonate.
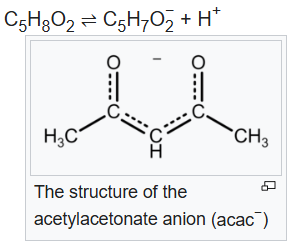
Acetylacetone is an organic compound with the chemical formula CH3−C(=O)−CH2−C(=O)−CH3. It is classified as a 1,3-diketone. It exists in equilibrium with a tautomer CH3−C(=O)−CH=C(−OH)−CH3.Acetylacetone is a weak acid. It forms the acetylacetonate anion
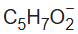
(commonly abbreviated acac−):
Dimethyl malonate is a diester derivative of malonic acid. It is a common reagent for organic synthesis used, for example, as a precursor for barbituric acid.
B is least acidic due to the +M effect of -OCH3 group. +I effect of the methyl group is present in A. +M effect dominates over the +I effect.