Correct option is A
In organic chemistry, a sigmatropic reaction is a pericyclic reaction wherein the net result is one sigma bond (σ-bond) is changed to another σ-bond in an intramolecular reaction. In this type of rearrangement reaction, a substituent moves from one part of a π-system to another part with simultaneous rearrangement of the π-system.
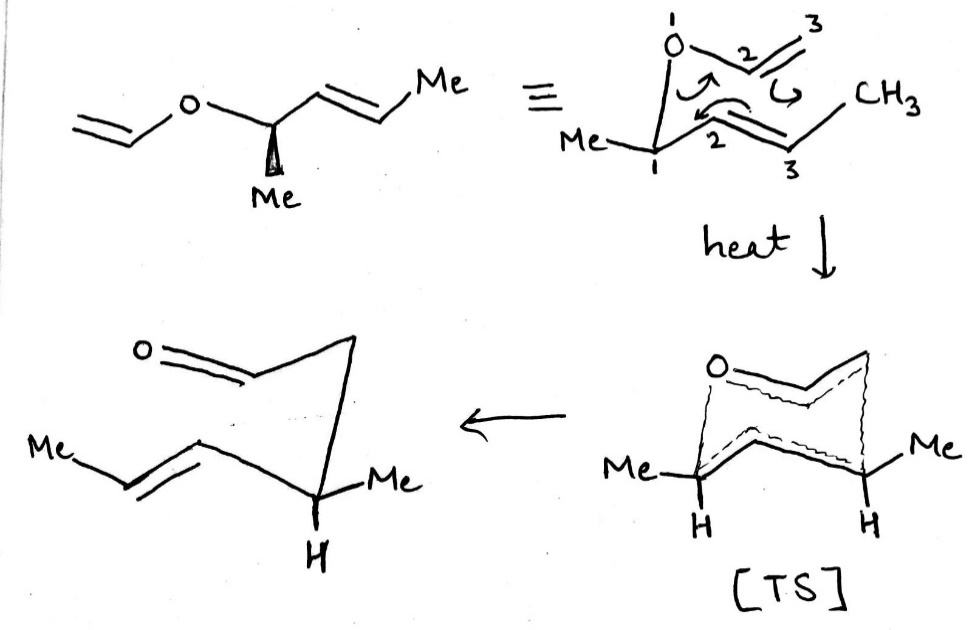
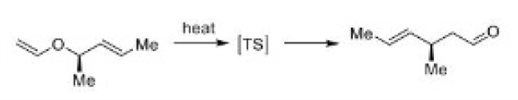
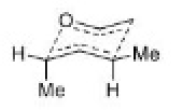
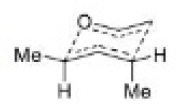
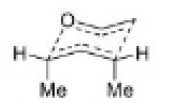
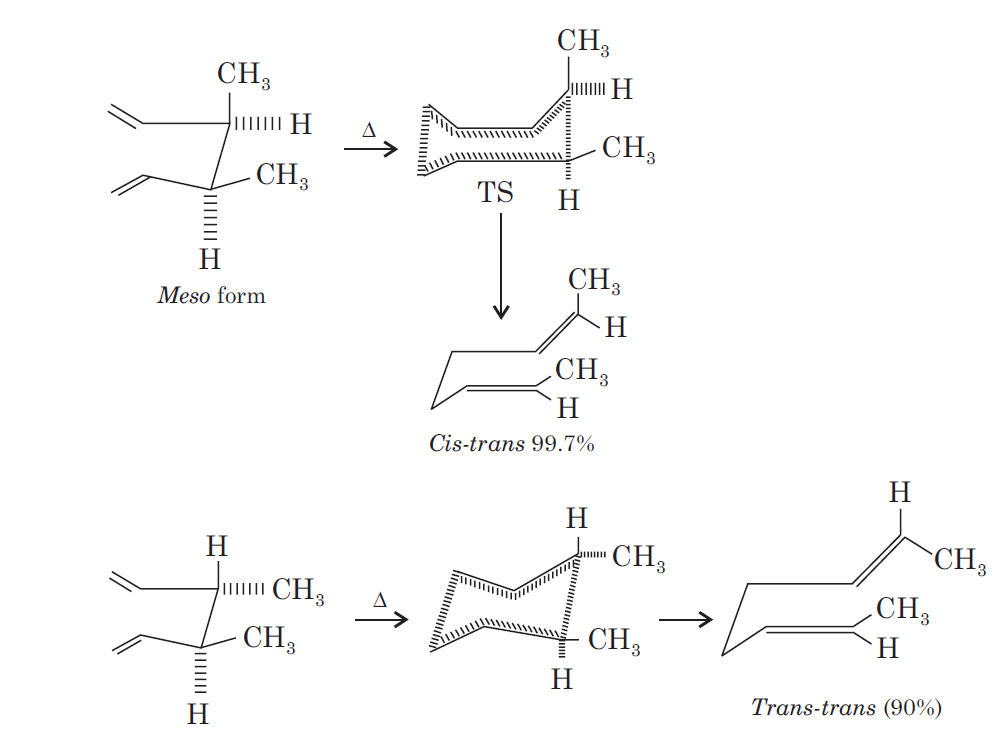
The most important sigmatropic rearrangement are the [3, 3] process involving carbon-carbon bond. The thermal rearrangement of 1, 5-dienes by [3, 3] sigmatropy is called Cope rearrangement.
This particular reaction is called a [3, 3] sigmatropic rearrangement because the new σ bond has a 3, 3 relationship to the old σ ( sigma) bond.
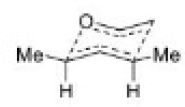
The Cope rearrangement usually proceeds through the chair like transition state. The stereochemical features of the reaction can usually be predicted and analysed on the basis of a chair transition state that minimizes steric interactions between substituents.
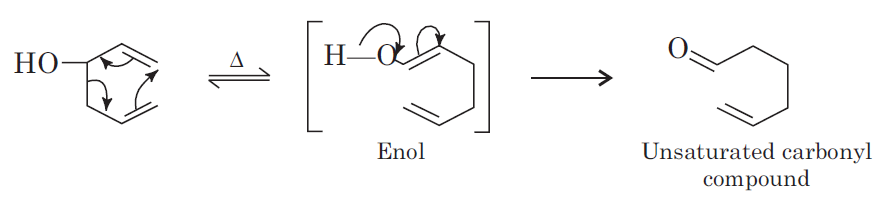
Preparation of Carbonyl Compounds from Cope Rearrangement
1, 5-Hexadiene-3-ol on heating undergoes Cope rearrangement with formation of unsaturated carbonyl compounds. Cope rearrangement given by such compounds is known as oxy-Cope rearrangement.