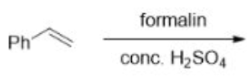Correct option is A
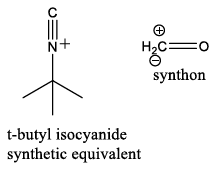
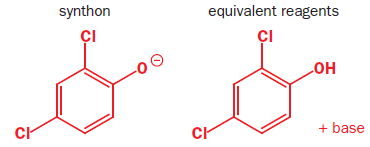
Synthetic equivalent: A reagent carrying out the function of a synthonwhich cannot itself be used, often because it is too unstable. Synthons are idealized fragments resulting from a disconnection. Synthons need to be replaced by reagents in a suggested synthesis.
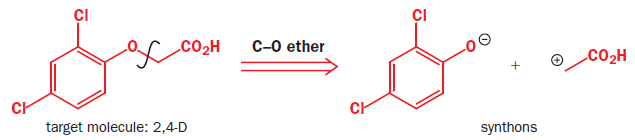
(2,4-dichlorophenoxyacetic acid)
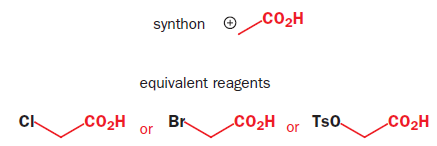
An isocyanide (also called isonitrile or carbylamine) is an organic compound with the functional group –N+≡C-. It is the isomer of the related nitrile (–C≡N), hence the prefix is isocyano. The organic fragment is connected to the isocyanide group through the nitrogen atom, not via the carbon. They are used as building blocks for the synthesis of other compounds.
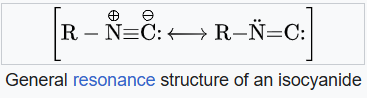
Isocyanides are described by two resonance structures, one with a triple bond between the nitrogen and the carbon and one with a double bond between. The π lone pair of the nitrogen stabilizes the structure and is responsible of the linearity of isocyanides, although the reactivity of isocyanides reflects some carbene character, at least in a formal sense. Thus, both resonance structures are useful representations. They are susceptible to polymerization.
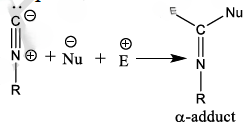
As opposed to most functional groups in organic chemistry, isocyanides react with both nucleophiles and electrophiles at the same carbon. This alpha–adduct forms the basis for all of the subsequent rearrangements and product forming steps.