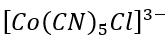Correct option is C
Replacement of a ligand from coordination sphere by OH- is called base hydrolysis. The most suitable mechanism has been given by Garrick which is called as SN1cB (substitution, nucleophilic, first order, conjugate base) mechanism.
The mechanism of Co (III) complexes containing amine ligands is shown below:
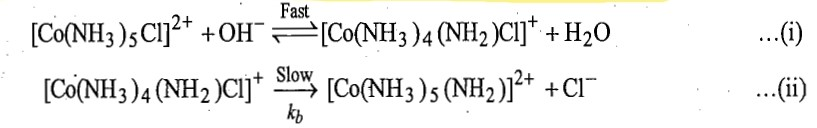

The first step involves the removal of proton from NH3 group by OH- in a rapid acid-base equilibrium forming amide complex ion of lower charge (a conjugate base of original amine complex). Then this conjugate base dissociates to loss Cl- in slow step [reaction (ii)] and finally it forms the hydroxo complex rapidly [reaction (iii)]. Thus, the second step is the rate determining step.
In amido complex (i.e., conjugate base) the NH2- ligand has the tendency to form π-bond by donating its lone pair of electrons to the metal cation, Co (III).
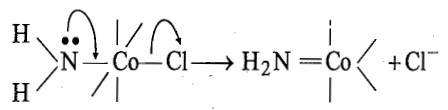
The formation of Co-N π-bond increases the electron density on cobalt which in turn repels the labile ligand, Cl- and the π-bond also stabilizes the five-coordinate intermediate. The donor π-bonding is strong evidence of an SN1 mechanism. Since the dissociation step which is the rate determining step uses the conjugate base of the original complex, therefore, it is called as SN1cB mechanism. It has been suggested that, the hydroxide ion abstract proton from the NH3 group which is trans to the leaving group, i.e., the halo group.
The rate determining step is dissociation of conjugate base [reaction (ii)] but since its concentration depends on the OH- ion concentration through equilibrium and the reaction rate is proportional to OH- ion concentration.
For base hydrolysis it is essential that there should be at least one ligand in a complex which have acidic protons.
The rate of hydrolysis of [Co (CN)5Cl]3- is independent of OH- concentration because of the absence of acidic proton.