Correct option is A
High Spin-Low Spin Equilibria
Octahedral complexes of d4, d5, d6and d7of first series transition metal ions can either be high spin or low spin depending on the magnitude of ligand field splitting Δo and pairing energy, P. When the magnitude of Δo has an intermediate value in such a way that the two complexes (LS and HS) have same energy, the two complexes can exist in equilibrium. The low spin and high spin complexes of a metal cation can be distinguished by measurement of either magnetic susceptibility or magnetic moments. For high spin complexes Δo < P and for low spin complexes Δo > P. If the difference in energy between Δo and P is small or negligible, the low spin and high spin can coexist in equilibrium.
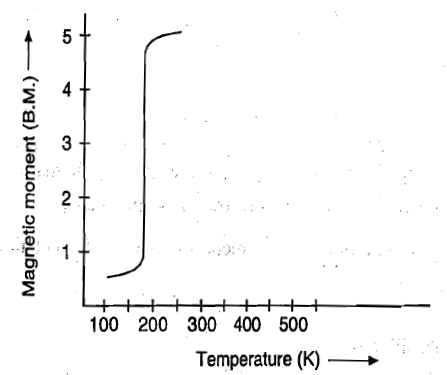
The choice between a low- and high-spin configuration for d4, d5, d6 and d7 complexes is not always unique and a spin crossover sometimes occurs. This may be initiated by a change in pressure (e.g. a low- to high-spin crossover for [Fe(CN)5(NH3)]3-at high pressure) or temperature (e.g. octahedral [Fe(phen)2(NCS-N)2], undergo low- to high-spin crossovers at 175K). The change in the value of effective magnetic moment which accompanies the spin crossover may be gradual or abrupt.
In addition to magnetic measurements, Mössbauer spectroscopy can be used to study spin-crossover transitions. Isomer shifts of iron complexes are sensitive not only to oxidation state but also to spin state. The Mössbauer spectra of [Fe{HC(3,5-Me2pz)3}2]I2 was studied over a temperature range from 295 to 4.2 K. Each spectrum is characterized by a ‘split peak’ which is described in terms of the isomer shift, and the quadrupole splitting, ΔEQ. At 295 K, the iron(II) centre is high-spin. On cooling, the complex undergoes a change to a low-spin state, and at 4.2 K, the transition is complete.
[Fe(phen)2(NCS)2]: At high temperature this complex exists as high spin with four unpaired electrons. When the temperature is decreased, there is sharp decrease in magnetic moment at 174 K and the complex becomes low spin.