Correct option is D
Transition metal dinitrogen complexes are coordination compounds that contain transition metals as ion centers the dinitrogen molecules (N2) as ligands. In terms of its bonding to transition metals, N2 is related to CO and acetylene as all three species have triple bonds. A variety of bonding modes have been characterized. Based on whether the N2 molecules are shared by two more metal centers, the complexes can be classified into mononuclear and bridging. Based on the geometric relationship between the N2 molecule and the metal center, the complexes can be classified into end-on or side-on modes. In the end-on bonding modes of transition metal-dinitrogen complexes, the N-N vector can be considered in line with the metal ion center, whereas in the side-on modes, the metal-ligand bond is known to be perpendicular to the N-N vector.
Mononuclear, end-on
This relationship is illustrated by the pair of complexes IrCl(CO)(PPh3)2 and IrCl(N2)(PPh3)2.In these mononuclear cases, N2 is both a σ-donor and a π-acceptor. The M-N-N bond angles are close to 180°. N2 is a weaker pi-acceptor than CO, reflecting the nature of the π* orbitals on CO vs N2. For this reason, few examples exist of complexes containing both CO and N2 ligand. For this reason, few examples exist of complexes containing both CO and N2 ligand.
Metal carbonyls are coordination complexes of transition metals with carbon monoxide ligands. Metal carbonyls are useful in organic synthesis and as catalysts or catalyst precursors in homogeneous catalysis, such as hydroformylation and Reppe chemistry.
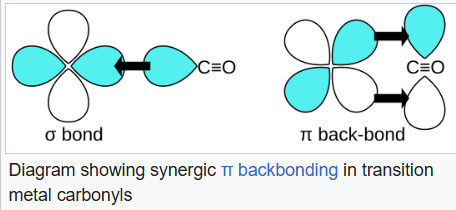
Carbon monoxide bonds to transition metals using "synergistic pi* back-bonding". The M–C bonding has three components, giving rise to a partial triple bond. A sigma (σ) bond arises from overlap of the nonbonding (or weakly anti-bonding) sp-hybridized electron pair on carbon with a blend of d-, s-, and p-orbitals on the metal. A pair of pi (π) bonds arises from overlap of filled d-orbitals on the metal with a pair of π*-antibonding orbitals projecting from the carbon atom of the CO. The latter kind of binding requires that the metal have d-electrons, and that the metal be in a relatively low oxidation state (0 or +1) which makes the back-donation of electron density favorable. As electrons from the metal fill the π-antibonding orbital of CO, they weaken the carbon–oxygen bond compared with free carbon monoxide, while the metal–carbon bond is strengthened.