Correct option is D
Orgel diagrams are particularly useful in the interpretation of spin allowed electronic transitions of tetrahedral and high spin octahedral complexes but not for low spin octahedral complexes. The spin allowed electronic transitions occur between the two energy states that have same spin multiplicity.

Combined Orgel diagram is shown in figure
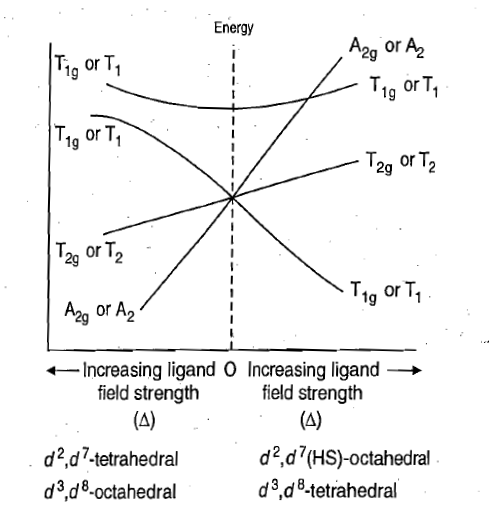
[Cr(en)3]3+:There are three possible spin allowed d-d transitions. It is a d3complex (Cr3+).
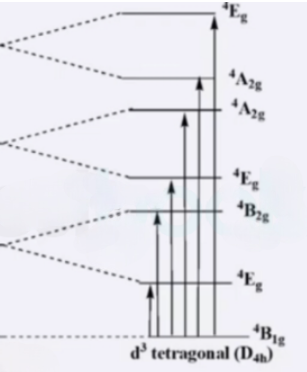
trans-[Cr(en)2F2]+: It has D4h symmetry. Six transitions are possible.