Correct option is B
Crystal field theory
This is an electrostatic model and simply uses the ligand electrons to create an electric field around the metal centre. Ligands are considered as point charges and there are no metal–ligand covalent interactions.
The octahedral crystal field
Consider a first row metal cation, Mn+, surrounded by six ligands placed on the Cartesian axes at the vertices of an octahedron. Each ligand is treated as a negative point charge and there is an electrostatic attraction between the metal ion and ligands. However, there is also a repulsive interaction between electrons in the d orbitals and the ligand point charges. If the electrostatic field (the crystal field) were spherical, then the energies of the five d orbitals would be raised (destabilized) by the same amount. However, since the dz2 and dx2-y2 atomic orbitals point directly at the ligands while the dxy, dyz and dxz atomic orbitals point between them, the dz2 and dx2-y2 atomic orbitals are destabilized to a greater extent than the dxy, dyz and dxz atomic orbitals. Thus, with respect to their energy in a spherical field (the barycentre, a kind of ‘centre of gravity’), the dz2 and dx2-y2 atomic orbitals are destabilized while the dxy, dyz and dxz atomic orbitals are stabilized.
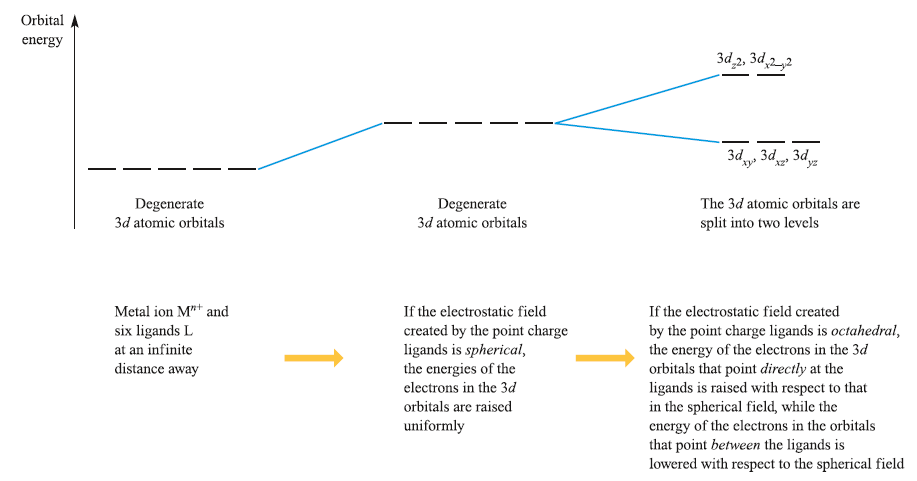
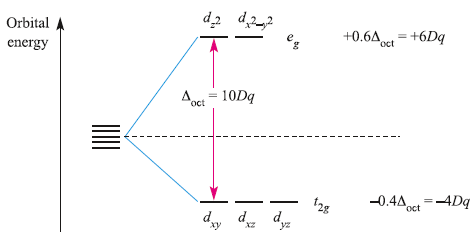
From the Oh character table, it can be deduced that the dz2 and dx2-y2 orbitals have eg symmetry, while the dxy, dyz and dxz orbitals possess t2g symmetry. The energy separation between them is Δoct (‘delta oct’) or 10Dq. The overall stabilization of the t2g orbitals equals the overall destabilization of the eg set. Thus, the two orbitals in the eg set are raised by 0.6Δoct with respect to the barycentre while the three in the t2g set are lowered by 0.4Δoct. The stabilization and destabilization of the t2g and eg sets, respectively, are given in terms of Δoct. The magnitude of Δoct is determined by the strength of the crystal field, the two extremes being called weak field and strong field.
Δoct (weak field) < Δoct (strong field)
For a ground state d4 ion, two arrangements are possible: the four electrons may occupy the t2g set with the configuration t2g4, or may singly occupy four d orbitals, t2g3eg1. Configuration 20.3 corresponds to a low-spin arrangement, and 20.4 to a high-spin case. The preferred configuration is that with the lower energy and depends on whether it is energetically preferable to pair the fourth electron or promote it to the eg level. Two terms contribute to the electron-pairing energy, P, which is the energy required to transform two electrons with parallel spin in different degenerate orbitals into spin-paired electrons in the same orbital:
the loss in the exchange energy which occurs upon pairing the electrons;
the coulombic repulsion between the spin-paired electrons.
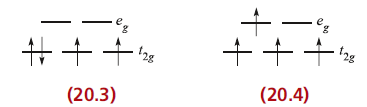
For high spin: Δoct< P and for low spin: Δoct > P
Electronic configuration of Ni is [Ar] 3d⁸4s². Ni2+=3d8. In both the high spin and low spin, the configuration will be t2g6eg2. Hence, the CFSE will be the same in both the cases.
Electronic configuration of Pt is [Xe] 4f¹⁴5d⁹6s¹. Pt2+=3d8. In both the high spin and low spin, the configuration will be t2g6eg2. Hence, the CFSE will be the same in both the cases.
Electronic configuration of Cr is [Ar] 3d⁵4s¹. Cr3+ =3d3. In both the high spin and low spin, the configuration will be t2g3eg0. Hence, the CFSE will be the same in both the cases.







