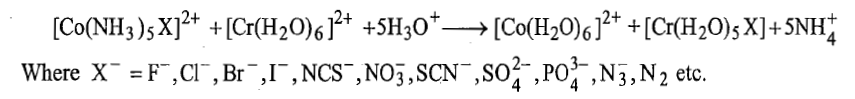Correct option is C
In inner sphere electron transfer reactions, the oxidant and reductant share a ligand in their coordination sphere to form a bridged complex, the electron is then transferred through the bridging ligand. An important reaction involving electron transfer by inner sphere mechanism is discussed below:
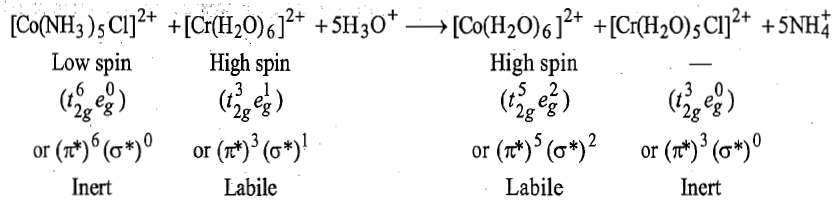
In this reaction Co3+is reduced by Cr2+and the bridging ligand Cl-is transferred from coordination sphere of cobalt to that of chromium.
i. is kinetically inert and Cl-is the bridging ligand.
ii. Electron transfer takes place from
Thus, electron transfer by inner sphere mechanism is fast (rate constant (k)=6×105Lmol-1s-1). The various steps of the mechanism of the reaction are shown below:
i. Chlorine atom of inert complex while remaining firmly attached to Co(III) replaces a water molecule from the labile to form a bridged intermediate.
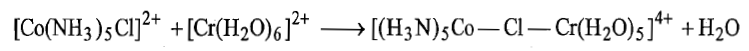
ii. Electron transfer takes place from Cr(II) to Co(III) through Cl bridge in a manner as electron flows between two electrodes through salt bridge. This step is the rate determining step. After transfer of electron from Cr2+ to Co3+, Co3+ is converted to Co2+ and the complex becomes high spin and labile. On the other hand, Cr2+ is converted to Cr3+ and the complex becomes inert.
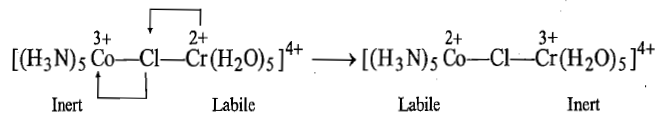
iii. Since Co2+is now labile and Cr3+is inert, so the bridged complex intermediate dissociates in such a way that chlorine atom remains attached with chromium.
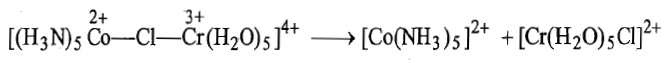
iv. In the last step, the five coordinate [Co(NH3)5]2+species picks up water molecule to form complex ion and this complex ion is hydrolyzed rapidly to give [Co(H2O)6]2+ion.
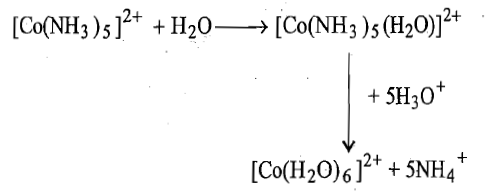
An evidence to support the inner sphere mechanism is that if the reaction is carried out in solution containing free isotopic chlorine (e.g.36Cl-), none of these labeled ions are found in the products. Conversely, if Cl- ion in [Co(NH3)5Cl]2+ is isotopically labeled with 36Cl, the labeled 36Cl is always transferred to Cr (III) coordination sphere. The reaction is first order with respect to oxidant and first order with respect to reductant i.e., overall reaction is of second order.
k is the second order rate constant.
The general reaction is given below: