Correct option is B
The orbital contribution is possible only when an orbital will transform into an equivalent orbital by rotation. The t2g orbitals (dxy, dyz and dzx) can be transformed into each other by rotating about an axis by 90°. The eg, orbitals (dx2-y2 and dz2) can not be transformed into one another because these have different shapes. Therefore, these orbitals have no orbital contribution. If all the t2g orbitals are singly occupied, then it is not possible to transform an orbital, say the dxy orbital into dyz or dzx orbital because they already contain an electron with same spin as the incoming electron. Similarly, it is impossible to transform a t2g orbital into others if all the t2g orbitals are doubly occupied. Thus, t2g orbitals with t2g3 and t2g6 configuration have no orbital contributions. The configurations other than t2g3 and t2g6make orbital contributions to the magnetic moments of octahedral complexes. Therefore, in octahedral complexes the following configurations make orbital contributions :
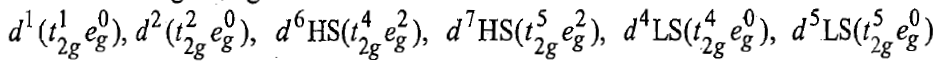
The following configuration in octahedral complexes of first series transition metal ions have no orbital contributions:
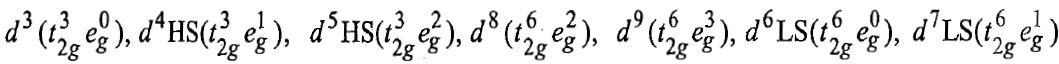
In a similar way, the tetrahedral complexes with the following configurations have orbital contributions:
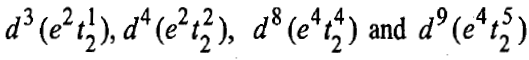
Option A: Co2+(d7):e4t23, Ni2+(d8): t2g6eg2
Option B: Ni2+(d8): e4t24, Co2+(d7): t2g5eg2
Option C: Co2+(d7): t2g5eg2, Cr3+(d3): t2g3eg0
Option D: Ni2+(d8): t2g6eg2, Cr2+(d4): e2t22







