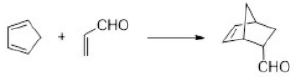
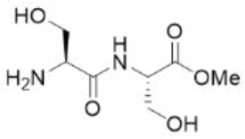
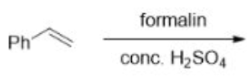Correct option is D
A solvent is a substance that dissolves a solute, resulting in a solution. A solvent is usually a liquid but can also be a solid, a gas, or a supercritical fluid. Water is a solvent for polar molecules, and the most common solvent used by living things; all the ions and proteins in a cell are dissolved in water within the cell.
Solvents can be broadly classified into two categories: polar and non-polar.
The polarity, dipole moment, polarizability and hydrogen bonding of a solvent determines what type of compounds it is able to dissolve and with what other solvents or liquid compounds it is miscible. Generally, polar solvents dissolve polar compounds best and non-polar solvents dissolve non-polar compounds best; hence "like dissolves like". Strongly polar compounds like sugars (e.g. sucrose) or ionic compounds, like inorganic salts (e.g. table salt) dissolve only in very polar solvents like water, while strongly non-polar compounds like oils or waxes dissolve only in very non-polar organic solvents like hexane. Similarly, water and hexane (or vinegar and vegetable oil) are not miscible with each other and will quickly separate into two layers even after being shaken well.
In the first reaction, the product and reactant are polar. Changing the medium will not particularly favor the product and thus the rate will not change much.
In the second reaction, the reactant is more polar than the product. The presence of polar medium favors the reactant side. The rate will not increase.
In the third reaction, reactant and product are not stable in polar medium. The rate will not increase.
In the fourth reaction, the product is ionic. The product is more stable in polar medium than the reactants. The formation of the product will be favored. The reaction will show an increase in rate.