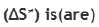Correct option is B
The change in entropy (ΔS) is a thermodynamic property that measures the difference in entropy between two states of a system. It quantifies how the entropy of a system changes as it undergoes a process from an initial state to a final state. ΔS can be calculated using the formula:
ΔS = ΔS final — Δ Sinitial
In the transition state of ester hydrolysis, the entropy change (ΔS) is usually negative. This is because transition states are generally characterized by a higher degree of organization and a lower degree of molecular randomness than reactants. Since bonds are being formed and broken in the transition state, the molecules are in a more disrupted and ordered state. This decrease in molecular randomness leads to a decrease in entropy, resulting in a negative ΔS for the transition state.
The activation entropy for the reaction between (CH3)3CCl (tertiary butyl chloride) and H2O typically tends to be negative. In this reaction, tertiary butyl chloride reacts with water in a nucleophilic substitution reaction. As the reaction proceeds, the transition state involves the simultaneous breaking of the carbon-chlorine bond and the formation of new bonds with the hydroxide ion (OH-). The reason for the negative activation entropy is that the transition state in this reaction tends to be more ordered and constrained compared to the reactants. As the carbon-chlorine bond is breaking and the carbon-oxygen bond is forming, the molecules in the transition state become more organized and less randomly oriented, leading to a decrease in entropy.
The reaction involving the molecule Me-O-O-Me (a peroxide compound) in the presence of heat, the activation entropy will be positive. When a reaction is initiated or catalyzed by the application of heat, the molecules gain thermal energy, which leads to increased molecular motion and disorder. This increase in molecular randomness and disorder results in a positive change in entropy (ΔS) for the transition state of the reaction.
Reaction B will also be spontaneous as the reaction intermediate is a tertiary carbocation formed which is very stable.