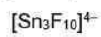Correct option is A
When hydrofluoric acid reacts with tin (II) oxide (SnO), the following reaction occurs:

H2SnF6 (P)
When HF reacts with tin (IV) chloride (SnCl4), a Lewis acid-base reaction occurs. SnCl4 is a Lewis acid because it can accept a pair of electron. HF is a Lewis base because it can donate a pair of electron. The reaction can be represented as follows:

When hydrogen hexafluorostannate (IV) (H2SnF6) reacts with sodium fluoride, it forms
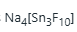
through a complexation reaction.
In the compound SnF4, the tin atom has +4 oxidation state and forms four strong bonds with fluorine atoms. It does not have readily available vacant orbitals to accept additional electron pairs, making it a weaker Lewis acid.