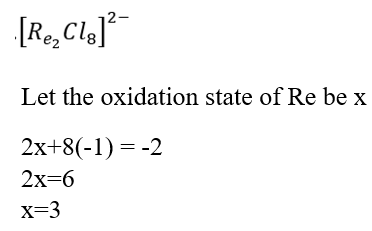Correct option is C
The [Re2Cl8]2- ion was the first inorganic compound containing a metal-metal quadruple bond.
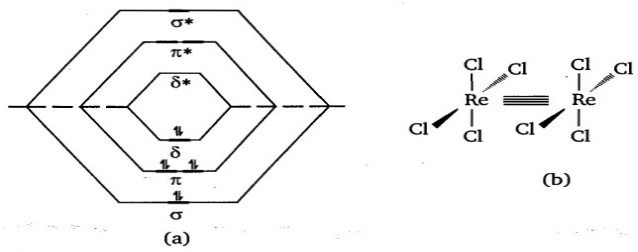
Each rhenium atom is bonded to four chlorine atoms that are almost in a square planar array. The dx2-y2 orbitals of the two metals are used for bonding to Cl- ligands. The Re-Cl bonds are formed by using dsp2-hybrid orbitals on both metals utilizing dx2-y2 orbitals. The metal dz2 and pz orbitals may hybridize to form two dp hybrid orbitals: one orbital directed towards the other rhenium atom and a second orbital directed in the opposite direction. The former hybrid orbital overlaps with the similar on the second Re atom to form a Re-Re σ bond whereas the second orbital forms a non-bonding orbital. The dxz and dyz orbitals of each metal atom overlap to form two π-bonds in xz and yz planes respectively. Finally, sideways overlap of dxy orbitals of the two metals form a δ bond.
The Re-Cl bonds are regarded as coordinate bonds between Re3+ions and the Cl- ligands. The eight d-electrons from two Re3+ions (four electrons from each Re3+ion) occupy a σ bonding, two π-bonding and one δ-bonding molecular orbitals to form the quadruple bond. Therefore, the electronic configuration in [Re2Cl8]2- ion will be σ2 π4 δ2.