Correct option is B
Butyl bromide reacts with AgNO2 to form nitrobutane. The N-O stretching vibrations in nitroalkanes occur near 1550 cm-1 (asymmetrical) and 1365 cm-1 (symmetrical).
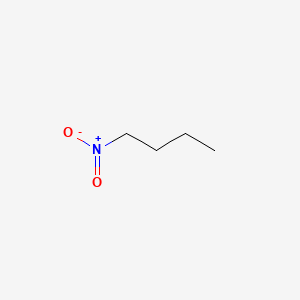
Oxidation state of oxygen is -2 while that of alkyl group is +1. Let the oxidation state of nitrogen be x. Thus, x-2-2+1=0, x=+3.
A formal charge, in the covalent view of chemical bonding, is the hypothetical charge assigned to an atom in a molecule, assuming that electrons in all chemical bonds are shared equally between atoms, regardless of relative electronegativity.
The formal charge of any atom in a molecule can be calculated by the following equation:
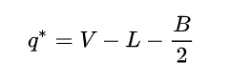
where V is the number of valence electrons of the neutral atom in isolation (in its ground state); L is the number of non-bonding valence electrons assigned to this atom in the Lewis structure of the molecule; and B is the total number of electrons shared in bonds with other atoms in the molecule.
On substituting the values, formal charge of nitrogen can be found as:5-0-8/2=1






