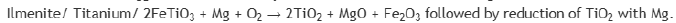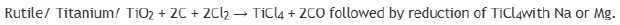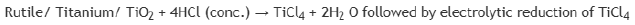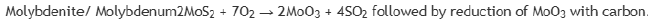Correct option is B
Metals are extracted from ores (found in earth's crust) which contain metals in non elemental form and other valuable minerals. Metals exists as their sulphides, oxides, carbonates, chlorides or silicates, etc. Metal in their elemental forms are extracted by reducing the metal ions present in their ore. Sulphide and carbonate ores are roasted and calcinated respectively to get metal oxides. The oxides of metal are then reduced by C or other suitable reducing agents or electrolysis.
Titanium exists as Ilmenite ore (FeTiO3) and rutile (TiO2) in earths crust. Impure titanium is extracted from it by treating them with Cl2 and C to get TiCl4 which can further be reduced by Na or Mg. The set of chemical process involved are:
Extraction of Mo: Molybdenum is found as its sulfide in the earth's crust. Firstly, the sulfide is converted to oxide by roasting in air at temperature higher than 500oC.