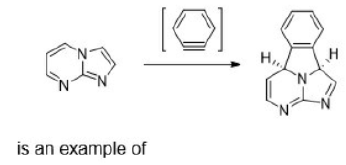
Correct option is C
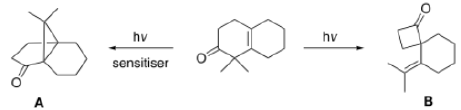
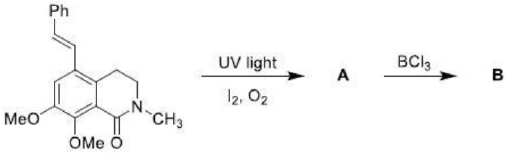
Product A: β, γ-unsaturation in a carbonyl compound promotes α-cleavage because of allylic stabilization of the radical produced. β, γ-unsaturated ketones, in addition to undergoing normal photochemical reactions of saturated ketones, undergo rearrangement reaction. The rearrangement is 1, 2-acyl shift (oxa-di-π-methane rearrangements) which occur from the lowest triplet state of n → π* transition.

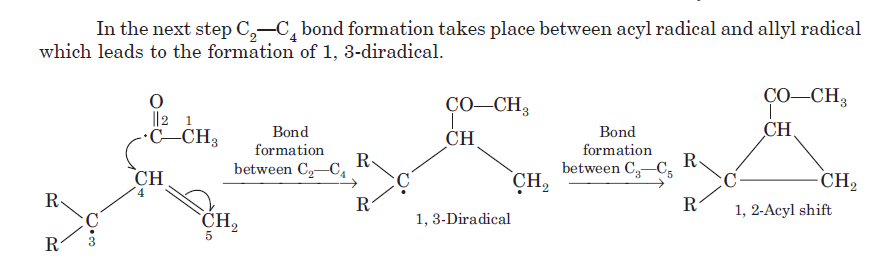
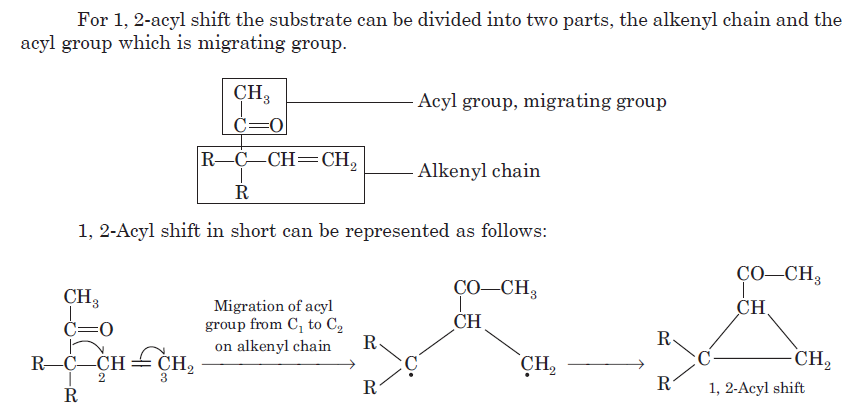
Since acyl group migrates from carbon-1 to carbon-2 of the alkenyl chain, the rearrangement due to this reason is known as 1, 2-acyl shift. 1, 2-Acyl shift given by β, γ-unsaturated ketones is known as oxa-di-π-methane rearrangement. 1, 2-acyl shift leads the formation of cyclopropane derivative.
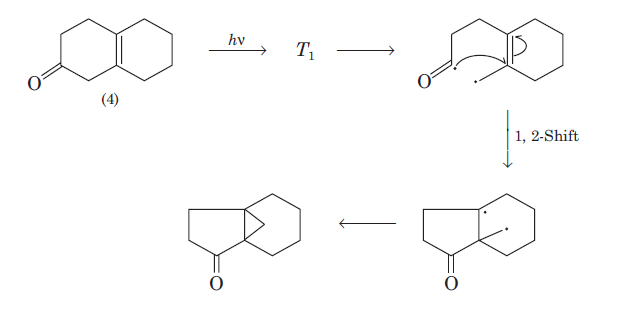
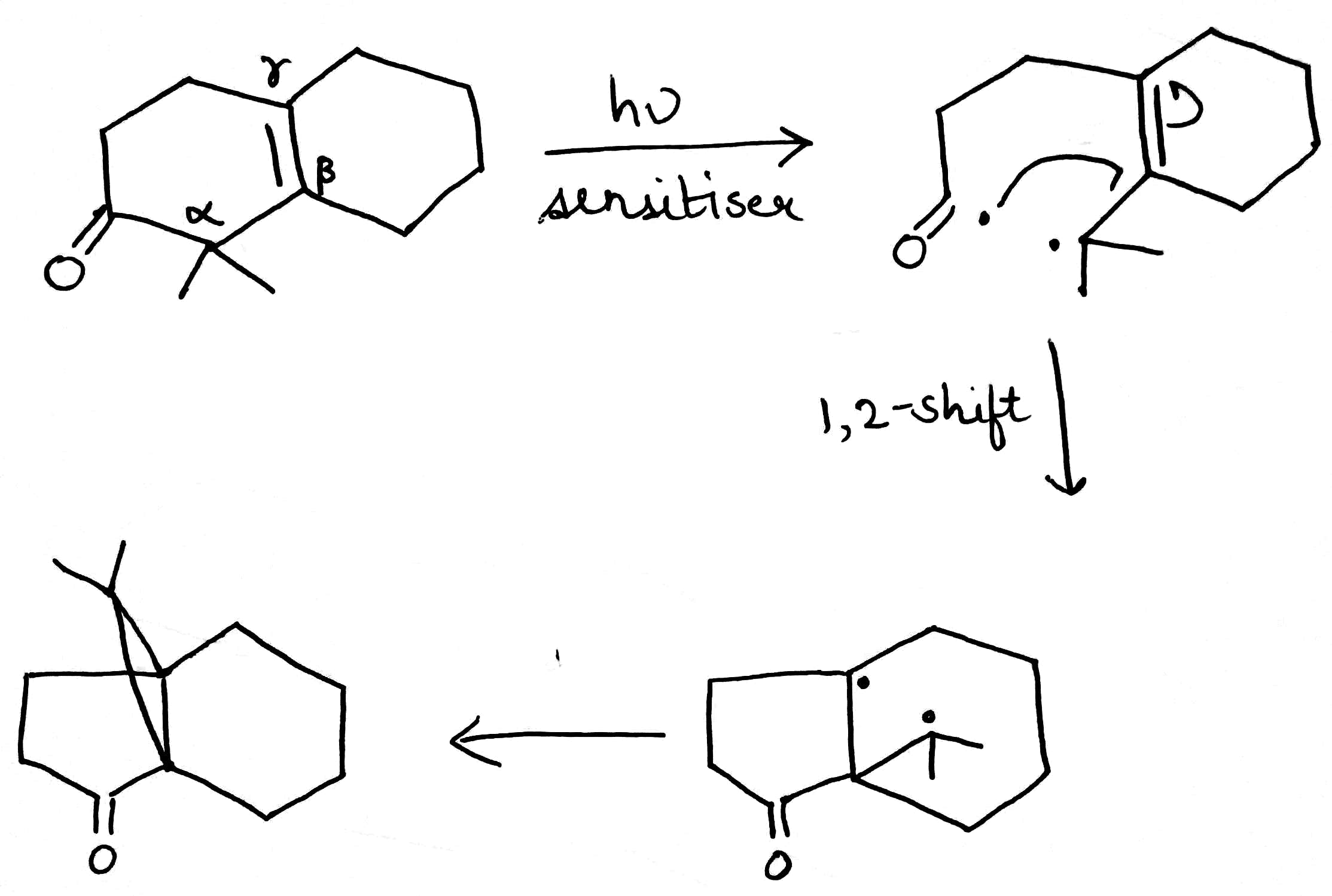
Oxa-di-π-methane (ODPM) rearrangement is generally much more efficient in rigid than in non-rigid systems.
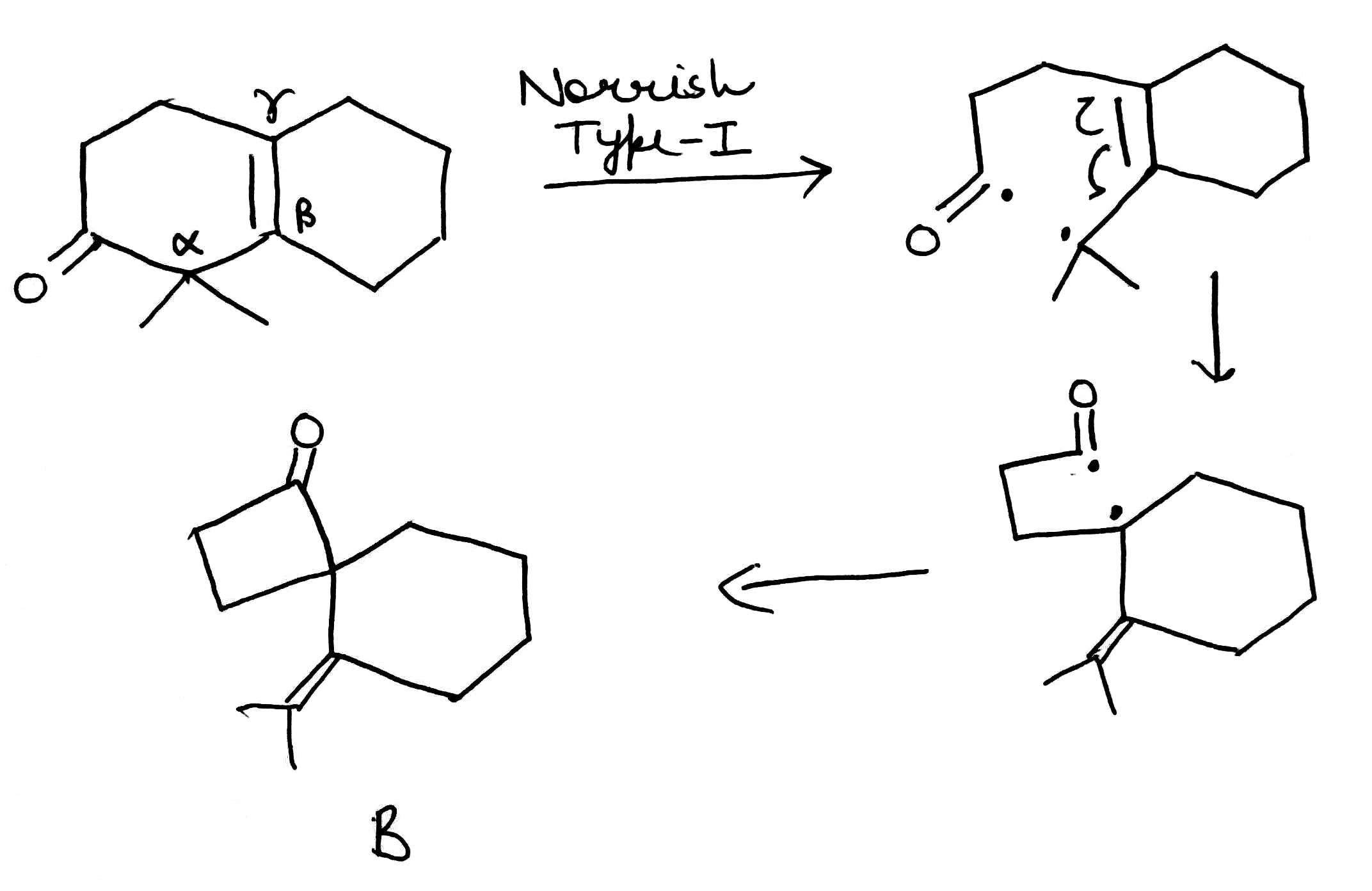
Product B: The Norrish type I reaction is the photochemical cleavage or homolysis of aldehydes and ketones into two free radical intermediates (α-scission). The carbonyl group accepts a photon and is excited to a photochemical singlet state. Through intersystem crossing the triplet state can be obtained. On cleavage of the α-carbon bond from either state, two radical fragments are obtained. The size and nature of these fragments depends upon the stability of the generated radicals.
Additional Knowledge
A Norrish type II reaction is the photochemical intramolecular abstraction of a γ-hydrogen (a hydrogen atom three carbon positions removed from the carbonyl group) by the excited carbonyl compound to produce a 1,4-biradical as a primary photoproduct.