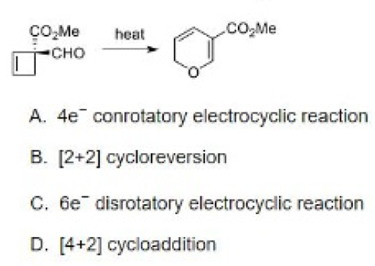
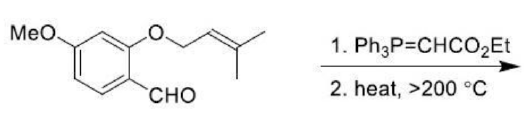
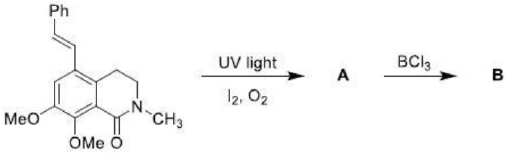
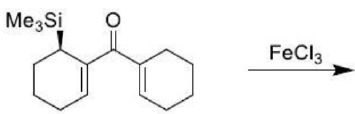
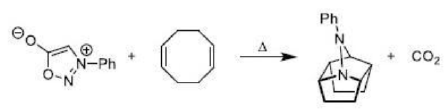
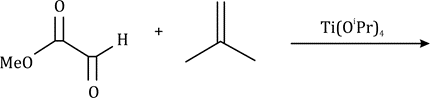
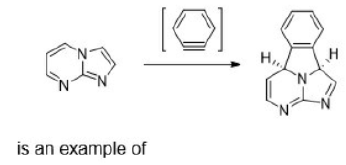
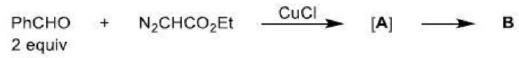Correct option is B
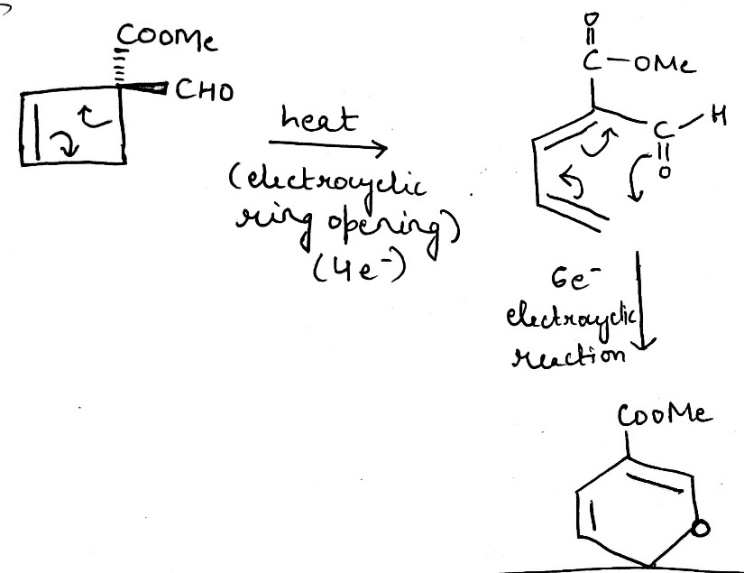
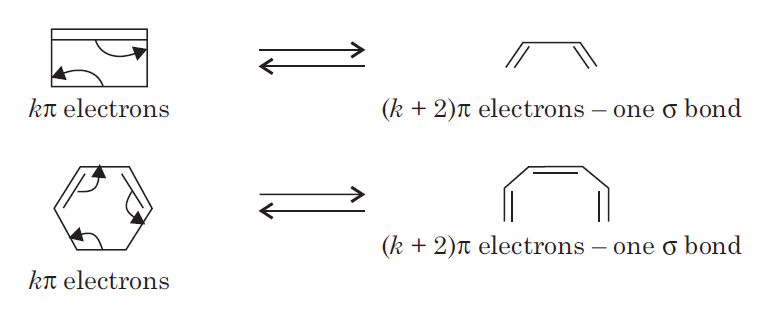
An electrocyclic reaction is the concerted interconversion of a conjugated polyene and a cycloalkene. Electrocyclic reactions are induced either thermally or photochemically.
All electrocyclic reactions are reversible reactions. Open-chain partner of the reaction is always conjugated system whereas cyclic partner may or may not contain conjugated system.
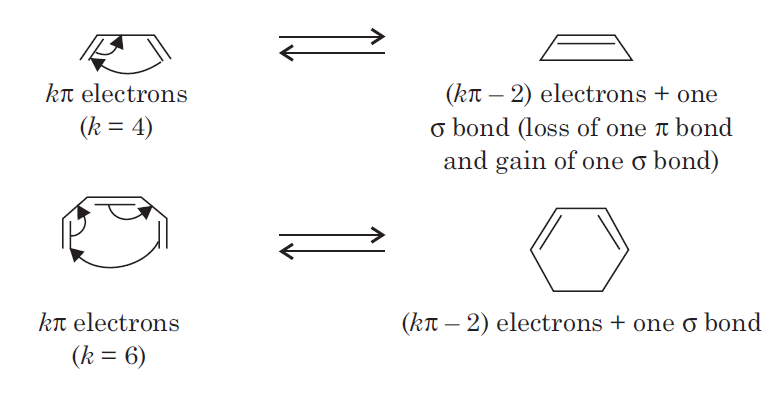
In electrocyclic reactions either a ring is formed with the generation of a new σ bond and the loss of a π bond ( i.e., gain of one σ bond and loss of one π bond) or ring is broken with the loss of one σ bond and gain of one π bond.
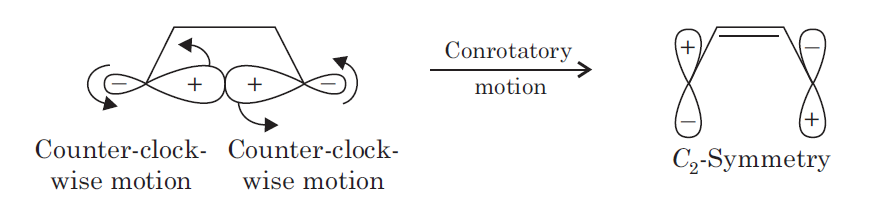
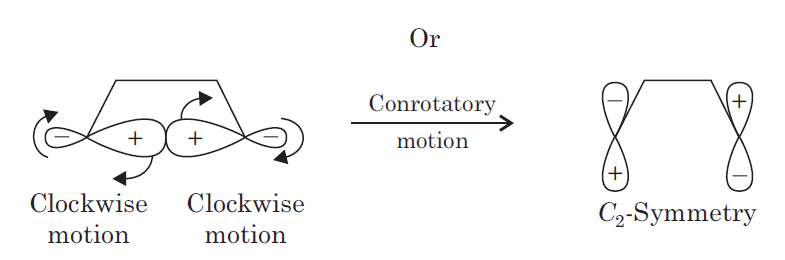
In organic chemistry, an electrocyclic reaction can either be classified as conrotatory or disrotatory based on the rotation at each end of the molecule. In conrotatory mode, both atomic orbitals of the end groups turn in the same direction (such as both atomic orbitals rotating clockwise or counter-clockwise). In disrotatory mode, the atomic orbitals of the end groups turn in opposite directions (one atomic orbital turns clockwise and the other counter-clockwise). The cis/trans geometry of the final product is directly decided by the difference between conrotation and disrotation.
The most common example of the ring opening reaction is the conversion of cyclobutene to 1, 3-butadiene.
This conversion can only be possible if a σ ( sigma) bond between C3 — C4 of cyclobutene must break during the course of the reaction.
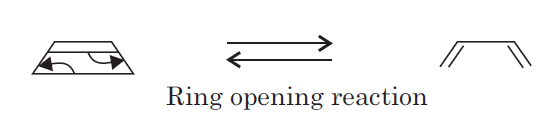
This σ ( sigma) bond may break in two ways. First, the two atomic orbital components of the σ ( sigma) bond may both rotate in the same direction, clockwise or counter-clockwise. This process is known as conrotatory motion.