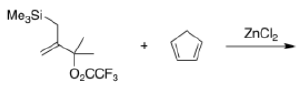
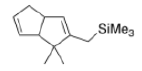
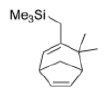
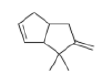
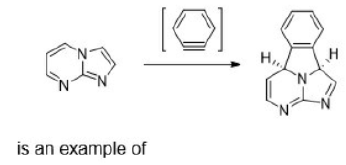
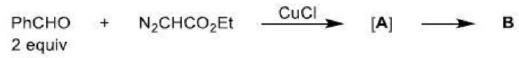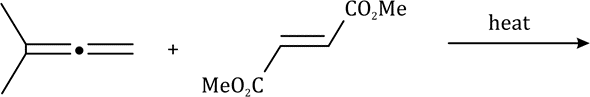Correct option is D
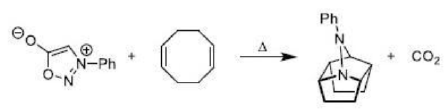
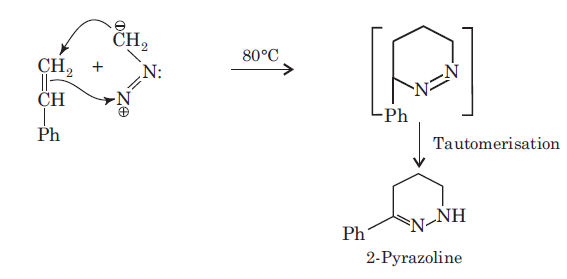
There is a large class of reactions, known as 1, 3-dipolar cycloaddition reactions. These reactions can be represented as in the given equation:
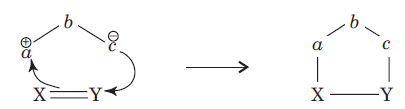
The species
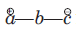
is called the 1, 3-dipolar molecule and X=Y is the dipolarophile. The 1,3-dipolar molecules are isoelectronic with the allyl carbanion and have four electrons in a π (pi) system encompassing the 1, 3-dipole. All 1, 3-dipoles contain 4π (pi) electrons in three parallel p orbitals of a, b and c.
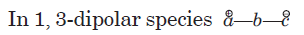
(i) a has six electrons in its outermost orbit. b has its complete octet having at least one lone pair of electrons. c has its complete octet having negative charge.
(ii) a may be carbon, oxygen or nitrogen, b may be nitrogen or oxygen, and c may be carbon, oxygen or nitrogen.
(iii) If b is nitrogen then it has single or double bond. If b is oxygen then it has single bond only.
The dipolarophiles (X=Y) are alkenes, alkynes, imines, nitriles and carbonyl compounds.
In most of the dipolar addition LUMO of the dipolarophile interacts with the HOMO of the 1, 3-dipole.
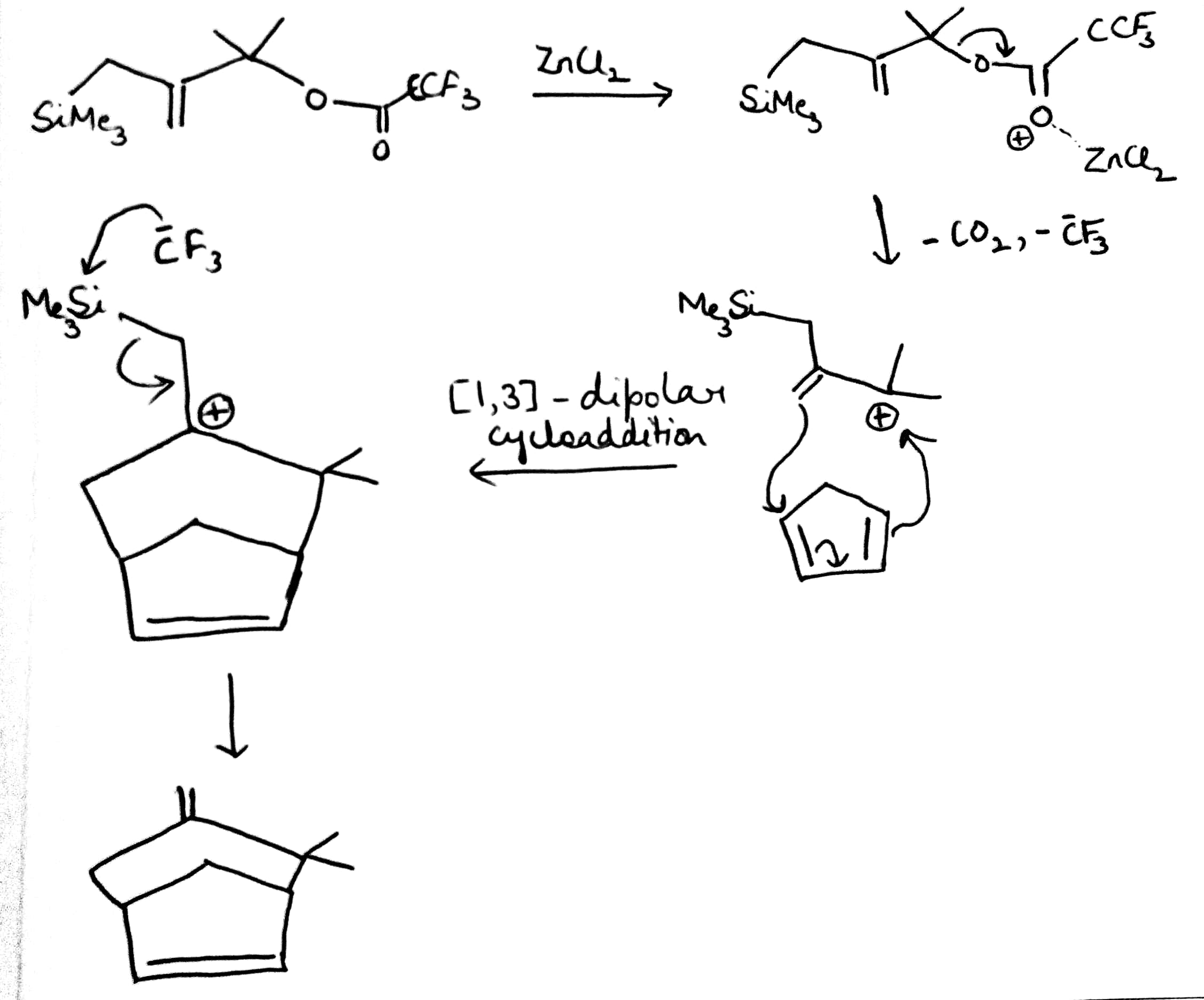
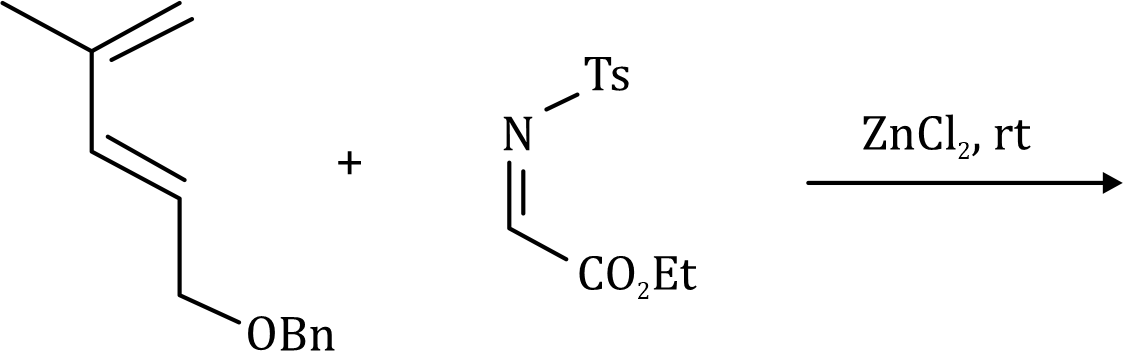
In the given reaction, ZnCl2 (a Lewis acid) will make the carbonyl carbon more electrophilic by interacting with the lone pair of oxygen atom.
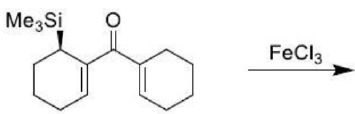
Trimethylsilyl and other silyl groups stabilize a positive charge on a β carbon and are lost very easily. They can be thought of as very reactive protons or ‘super protons’.