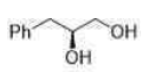
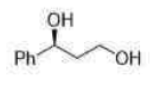
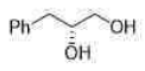
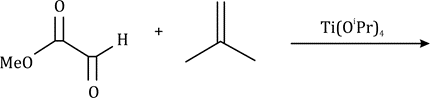
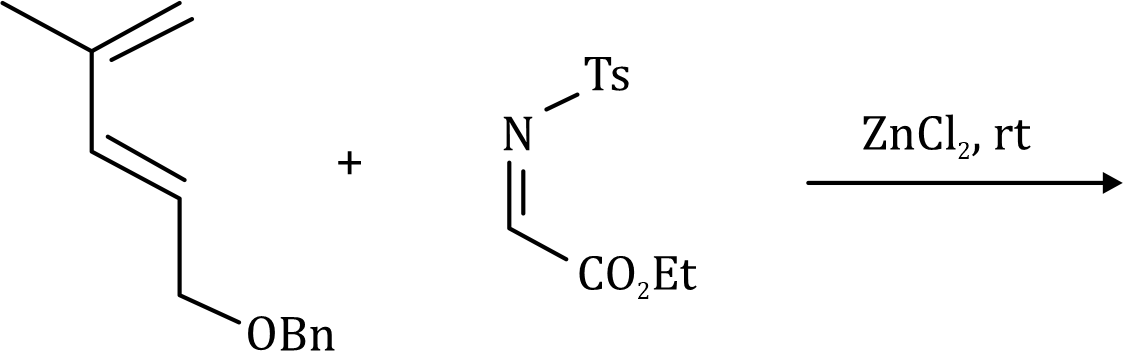
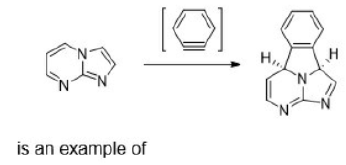
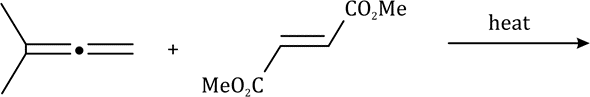Correct option is C
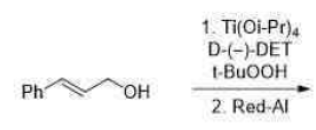
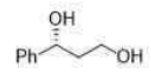
The Sharpless asymmetric epoxidation reaction is often used as a key step in synthetic protocols involving the synthesis of natural products such as terpenes, carbohydrates, insect pheromones, and pharmaceutical products. The SAE reaction is characterized by its simplicity and reliability. The epoxides are obtained with predictable absolute configuration and in high enantiomeric excess (ee). Moreover, 2,3-epoxy alcohols serve as versatile intermediates for a host of stereospecific transformations.
Prochiral allylic alcohols are epoxidized in the presence of t-BuOOH, Ti(i-OPr)4, and (+)-or (-)-diethy1 tartrate (DET) with high regio- and stereoselectivity to produce the corresponding optically active epoxides.
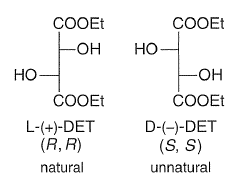
Diethyl tartrate, the allylic alcohol, and the oxidant t-BuOOH displace the isopropoxide groups on titanium to form the active Ti-catalyst in a complex ligand exchange pathway. From structural and kinetic studies, Sharpless proposed that oxygen transfer occurs from a dimeric complex that has one tartaric ester moiety per titanium atom.
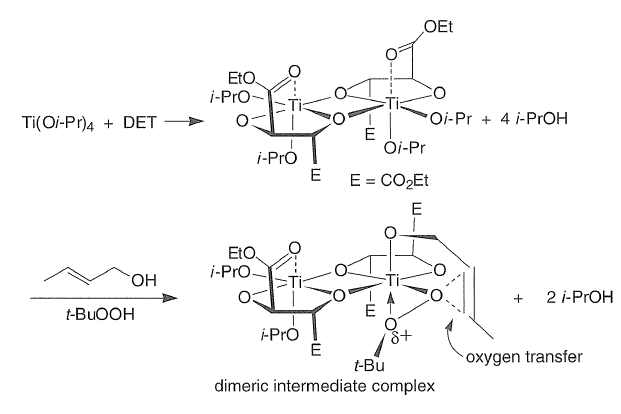
The absolute configuration of the epoxy alcohol is predictable using the model depicted below:
In case of (-)-DET, when the OH group in the allylic alcohol is on the right side then epoxide will form from below the plane(dash). On the other hand, when the OH group in the allylic alcohol is on the left side then epoxide will form from above the plane(wedge).
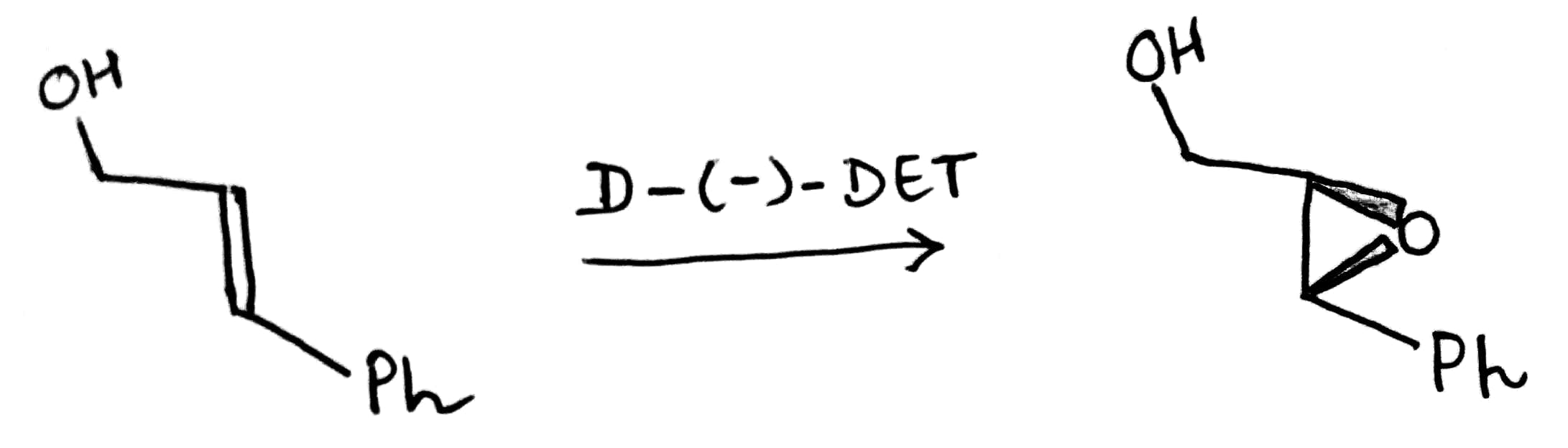
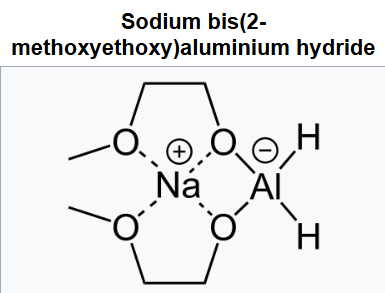
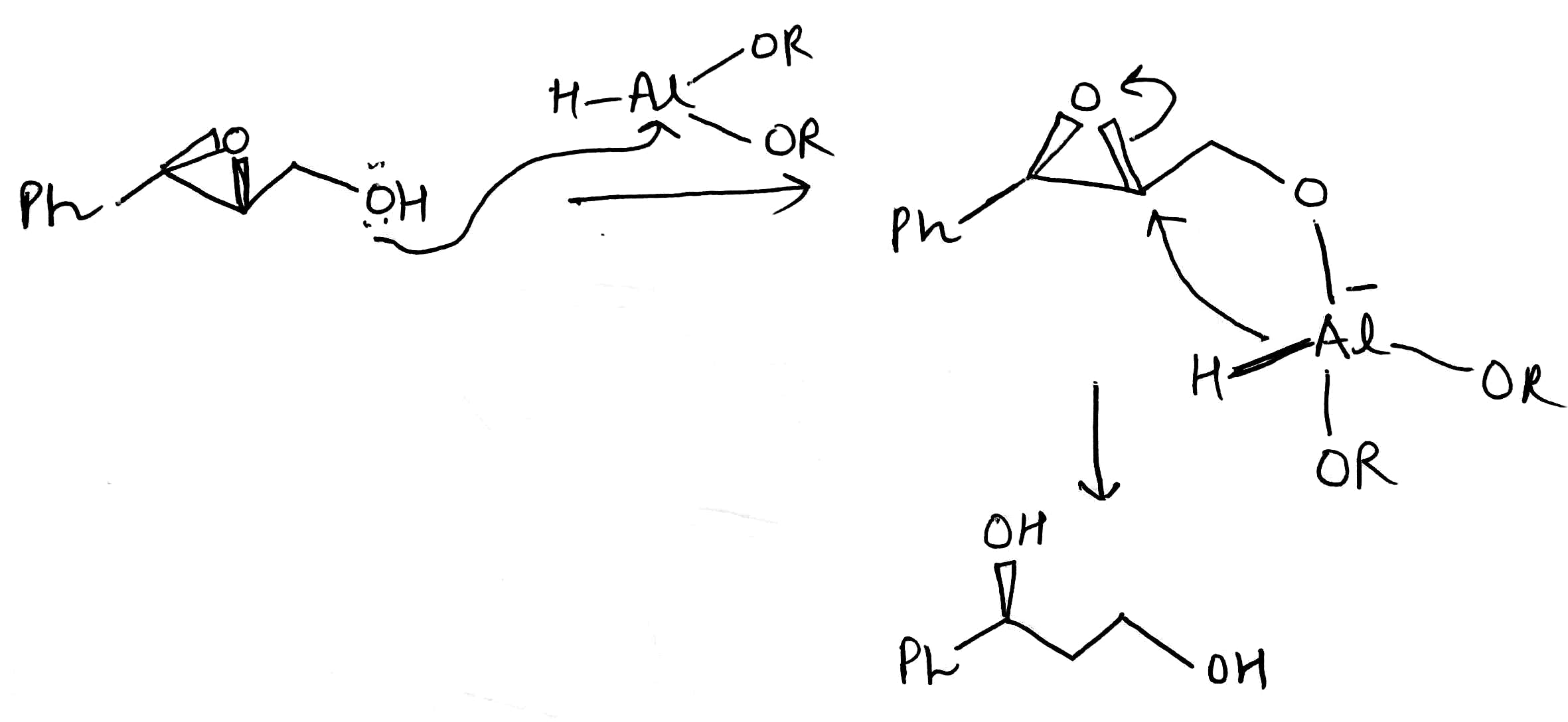
Sodium bis(2-methoxyethoxy)aluminium hydride (SMEAH; trade names Red-Al, Synhydrid, Vitride) is a hydride reductant with the formula NaAlH2(OCH2CH2OCH3)2. The trade name Red-Al refers to its being a reducing aluminium compound. It is used predominantly as a reducing agent in organic synthesis. The compound features a tetrahedral aluminium center attached to two hydride and two alkoxide groups, the latter derived from 2-methoxyethanol. SMEAH is a versatile hydride reducing agent. It readily converts epoxides, aldehydes, ketones, carboxylic acids, esters, acyl halides, and anhydrides to the corresponding alcohols.