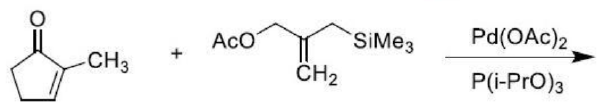
Correct option is A
Trimethylenemethane can be used to form a wide range of Five-membered rings such as cyclopentane, and cyclopentenes via [3 + 2] cycloaddition reaction, catalyzed by palladium(0) complexes. The trimethylenemethane unit in these reactions is generally derived from the 2-
[(trimethylsilyl)methyl]-2-propen-1-yl acetate, which is at the same time an allyl silane and an allylic acetate. Thus it is a weak nucleophile and an electrophile in the presence of palladium(0). Formation of the palladium -allyl complex is followed by the removal of the trimethylsilyl group by nucleophilic attack of the resulting acetate ion. This produces a zwitterionic palladium complex that can undergo the cycloaddition reaction as follows:
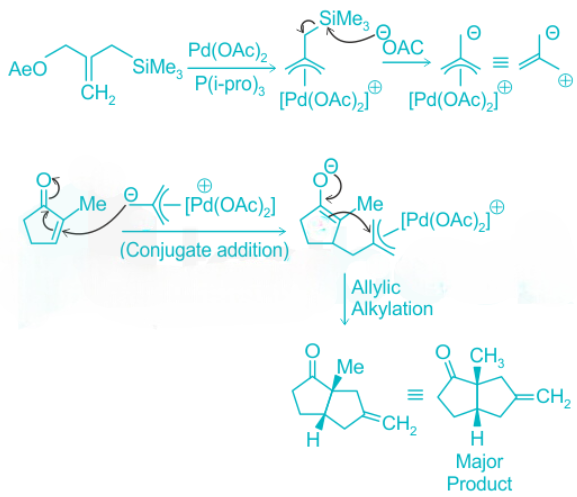
From the above reaction we can see that in the first step of the reaction, it forms a zwitterionic palladium complex. In the next step, the zwitterionic palladium complex undergoes a conjugate addition with the cyclopropane derivative followed by an attack of the resulting enolate on the t-allyl palladium unit to form a new five-membered ring having an Exo methylene group.