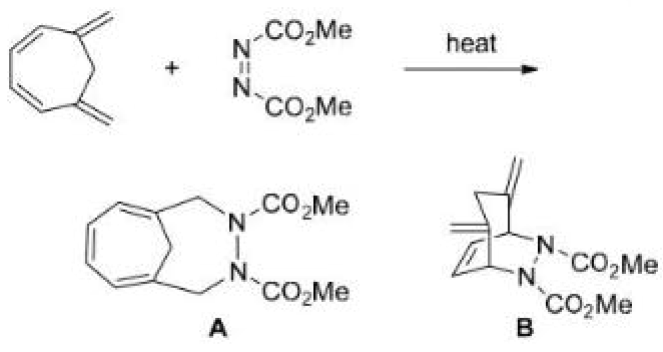
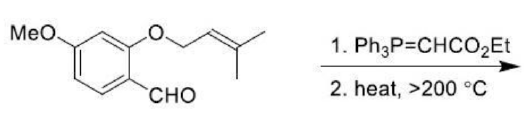
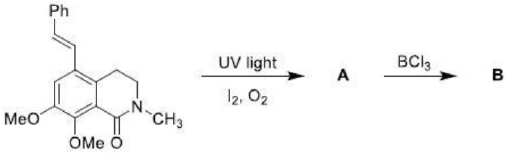
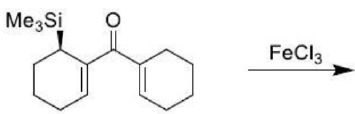
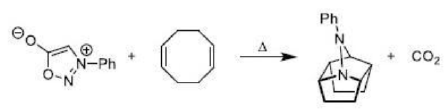
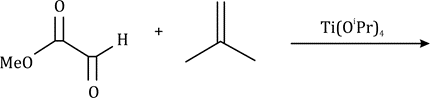
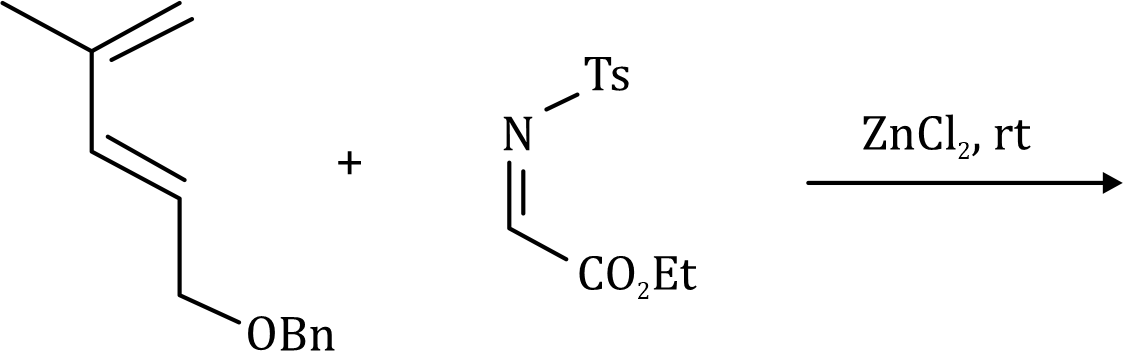
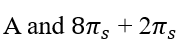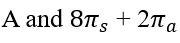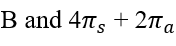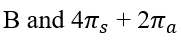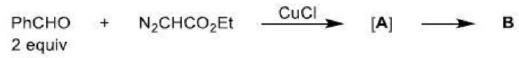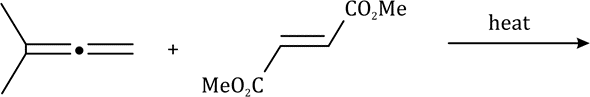Correct option is A
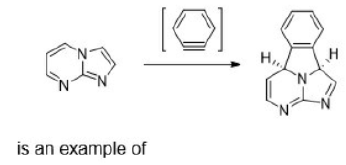
A cycloaddition is a chemical reaction in which "two or more unsaturated molecules (or parts of the same molecule) combine with the formation of a cyclic adduct in which there is a net reduction of the bond multiplicity". The resulting reaction is a cyclization reaction.
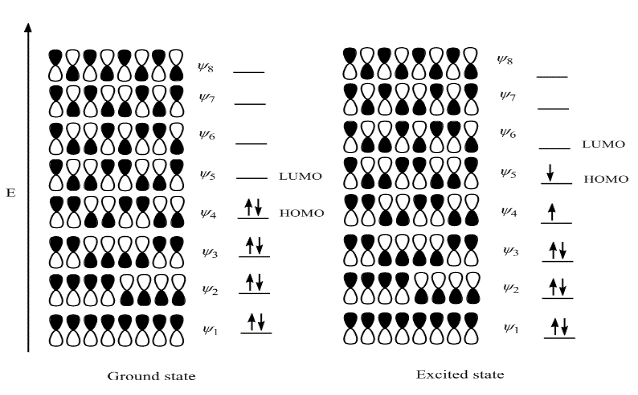
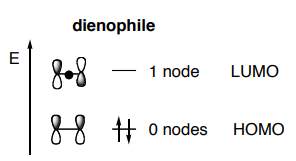
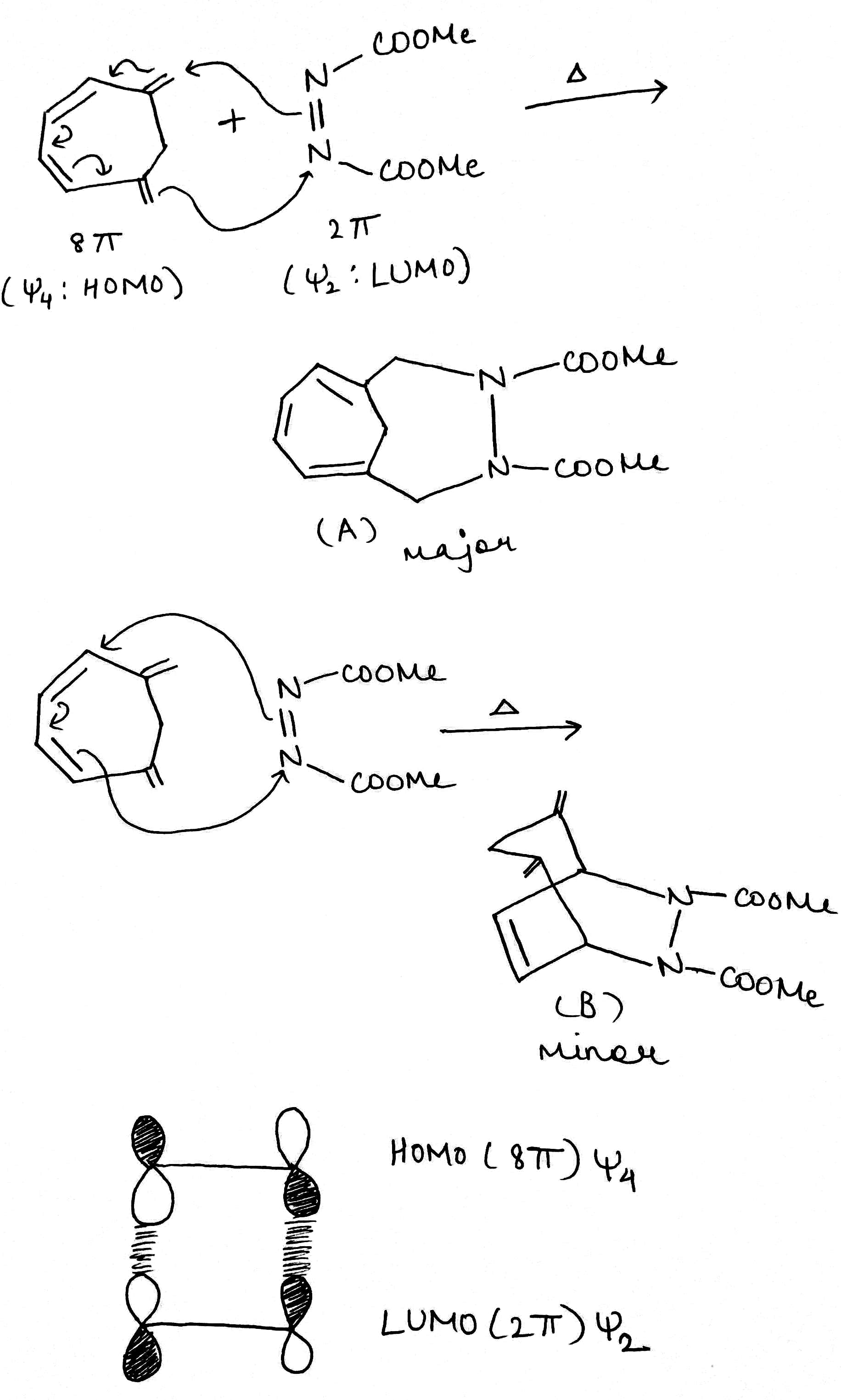
Thermal cycloadditions are those cycloadditions where the reactants are in the ground electronic state. They usually have (4 n + 2) π electrons participating in the starting material, for some integer n. These reactions occur for reasons of orbital symmetry in a suprafacial-suprafacial ( syn/ syn stereochemistry) in most cases. Very few examples of antarafacial-antarafacial ( anti/ anti stereochemistry) reactions have also been reported.
The relationship is antarafacial when opposite faces of the π system or isolated orbital are involved in the process (think anti). For a σ bond, it corresponds to involvement of one "interior" lobe and one "exterior" lobe of the bond. The relationship is suprafacial when the same face of the π system or isolated orbital are involved in the process (think syn). For a σ bond, it corresponds to involvement of two "interior" lobes or two "exterior" lobes of the bond.