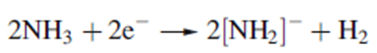Correct option is A
The common oxidation state for all the lanthanoids is Ln (III), which makes separation difficult, although cerium, which can be oxidized to Ce (IV), and europium, which can be reduced to Eu (II), are separable from the other lanthanoids by exploiting their redox chemistry.
The next most common of the atypical oxidation states is Eu2+ (f7), and there are a number of stable Eu2+ compounds, including EuI2, EuSO4, and EuCO3, and solutions of this ion are stable.
Eu forms blue solution in liquid NH3 due to the following reaction:
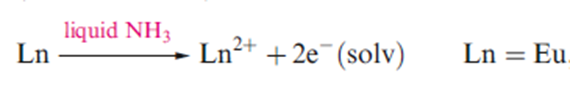
Information Booster
All of the group 1 metals and the group 2 metals Ca, Sr and Ba dissolve in liquid NH3 to give metastable solutions from which the group 1 metals can be recovered unchanged. The group 2 metals are recoverable as solids of composition
Dilute solutions of the metals are bright blue, the color arising from the short wavelength tail of a broad and intense absorption band in the infrared region of the spectrum. The electronic spectra in the visible region of solutions of all the s-block metals are the same, indicating the presence of a species common to all the solutions: this is the solvated electron.
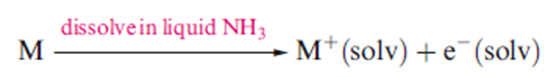
Each dilute solution of metal in liquid NH3 occupies a volume greater than the sum of the volumes of the metal plus solvent. These data suggest that the electrons occupy cavities of radius 300–400 pm. Very dilute solutions of the metals are paramagnetic, and the magnetic susceptibility corresponds to that calculated for the presence of one free electron per metal atom.
As the concentration of a solution of an s-block metal in liquid NH3 increases, the molar conductivity initially decreases, reaching a minimum at ~0.05 mol dm-3. Thereafter, the molar conductivity increases, and in saturated solutions, it is comparable with that of the metal itself. Such saturated solutions are no longer blue and paramagnetic, but are bronze and diamagnetic.
However, in order to explain why the magnetic susceptibilities of solutions decrease as the concentration increases, it is necessary to invoke equilibria at higher concentrations.
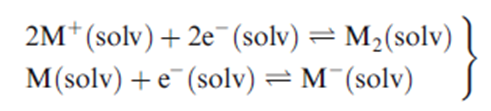
Hydrogen/deuterium isotopic substitution coupled with neutron diffraction studies confirm that the addition of an alkali metal to liquid NH3 disrupts the hydrogen bonding present in the solvent. In a saturated lithium–ammonia solution (21 mole percent metal), no hydrogen bonding remains between NH3 molecules. Saturated Li–NH3 solutions contain tetrahedrally coordinated Li, whereas saturated K–NH3 solutions contain octahedrally coordinated K.
The blue solutions of alkali metals in liquid NH3 decompose very slowly, liberating H2 as the solvent is reduced.