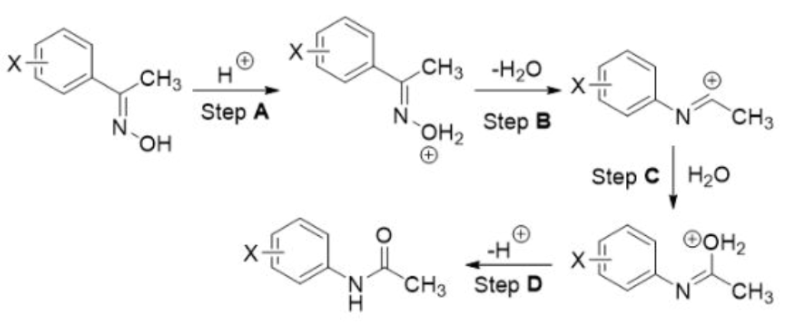
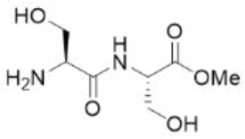
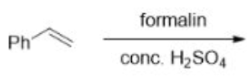Correct option is A
In organic chemistry, the Hammett equation describes a linear free-energy relationship relating reaction rates and equilibrium constants for many reactions involving benzoic acid derivatives with meta- and para-substituents to each other with just two parameters: a substituent constant and a reaction constant.
The basic idea is that for any two reactions with two aromatic reactants only differing in the type of substituent, the change in free energy of activation is proportional to the change in Gibbs free energy.
The basic equation is:
where

relating the equilibrium constant, K, for a given equilibrium reaction with substituent R and the reference constant K0 when R is a hydrogen atom to the substituent constant σ which depends only on the specific substituent R and the reaction rate constant ρ which depends only on the type of reaction but not on the substituent used.