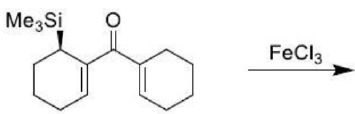
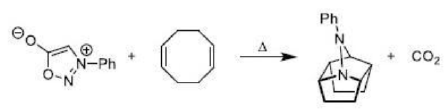
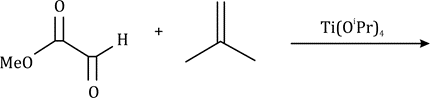
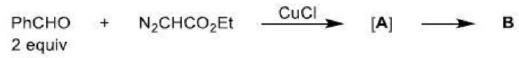Correct option is B
The Norrish type I reaction is the photochemical cleavage or homolysis of aldehydes and ketones into two free radical intermediates (α-scission). The carbonyl group accepts a photon and is excited to a photochemical singlet state. Through intersystem crossing the triplet state can be obtained. On cleavage of the α-carbon bond from either state, two radical fragments are obtained. The size and nature of these fragments depends upon the stability of the generated radicals.

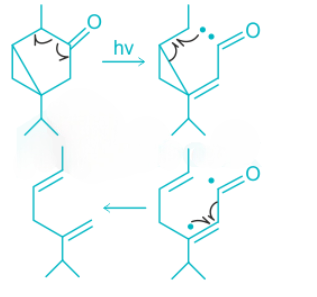
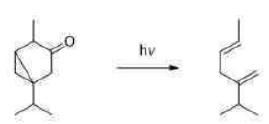
In the above reaction, alpha cleavage occurs in the presence of light. Alpha cleavage is followed by fragmentation of cyclopropane ring and release of CO.
Additional Knowledge
A Norrish type II reaction is the photochemical intramolecular abstraction of a γ-hydrogen (a hydrogen atom three carbon positions removed from the carbonyl group) by the excited carbonyl compound to produce a 1,4-biradical as a primary photoproduct.
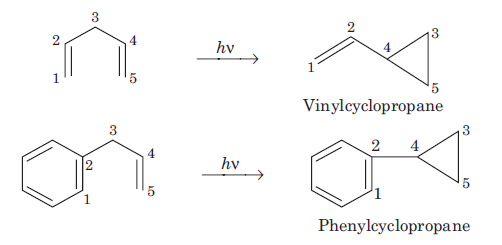
The most remarkable photochemical reaction of nonconjugated dienes is the di-π-methane rearrangement. This rearrangement is given by dienes having π (pi) system separated by an sp3 hybridized carbon atom i.e., 1, 4-pentadienes. Irradiation of the substrate induces rearrangement to a vinylcyclopropane or phenylcyclopropane derivative.