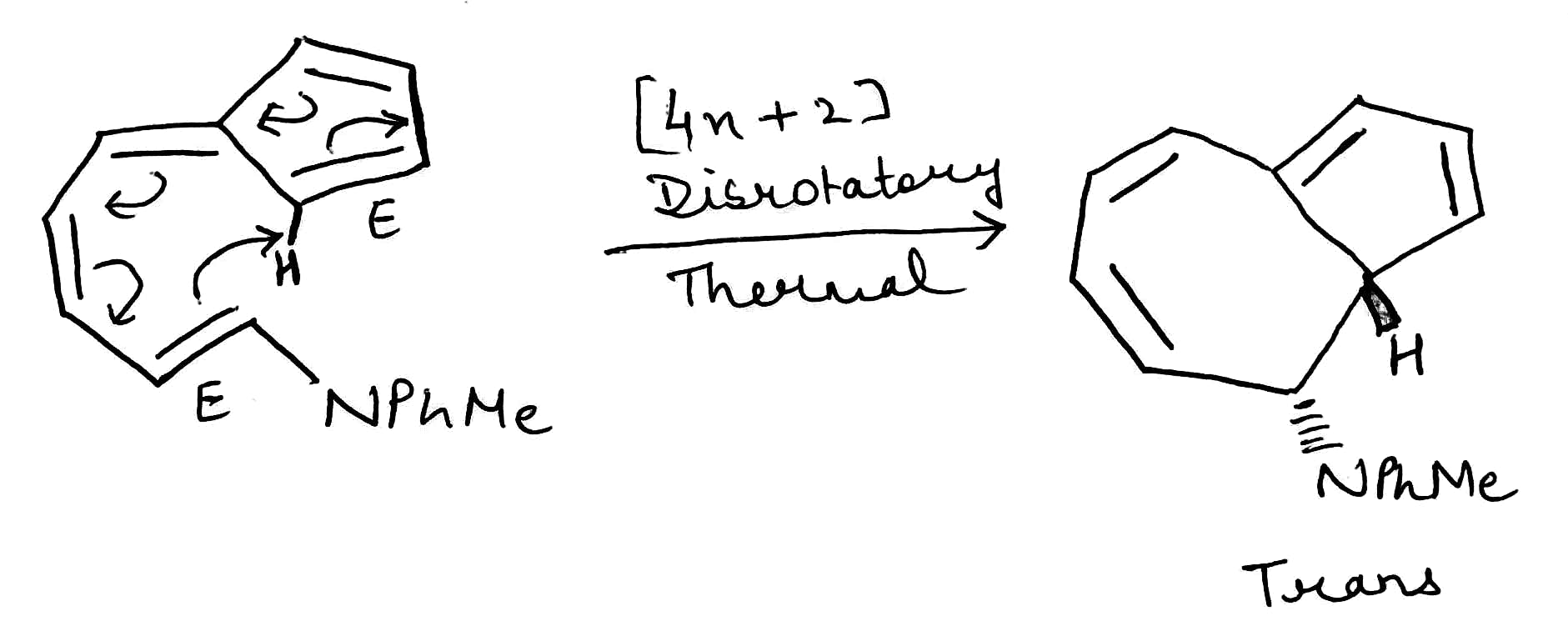
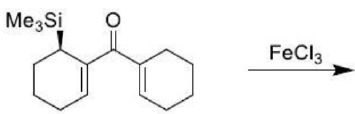
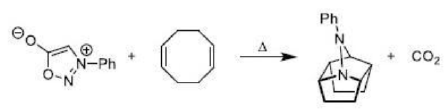
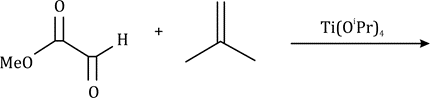
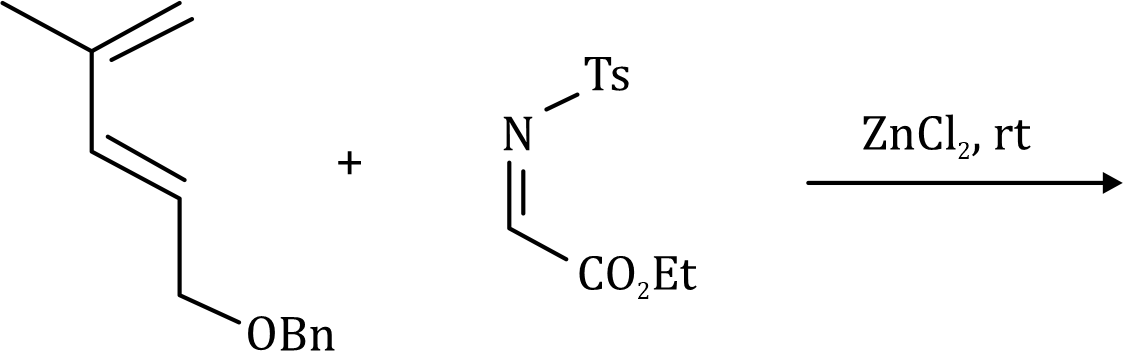
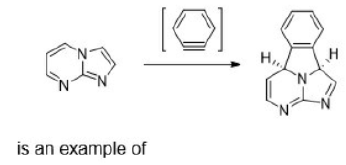
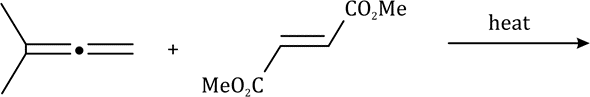Correct option is C
An electrocyclic reaction is the formation of a new sigma bond across the ends of a conjugated polyene or the reverse. These reactions are usually categorized by the following criteria:
Reactions can be either photochemical or thermal.
Reactions can be either ring-opening or ring-closing (electrocyclization).
Depending on the type of reaction (photochemical or thermal) and the number of pi electrons, the reaction can happen through either a conrotatory or disrotatory mechanism.
The type of rotation determines whether the cis or trans isomer of the product will be formed.
In conrotatory reactions the two groups rotate in the same way: both clockwise or both anticlockwise.
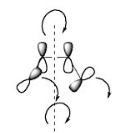
In disrotatory reactions, one group rotates clockwise and one anticlockwise.
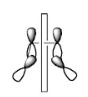
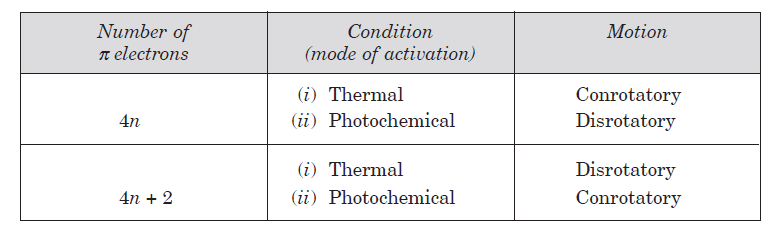
The given system contains 4(2)+2=10 pi electrons.