Correct option is D
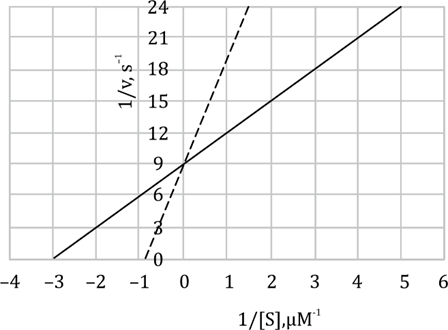
To solve this question, we need to calculate Kₘ and Kₛ (Michaelis constant and the rate constant for the enzyme-substrate complex dissociation, respectively) using the given values of the rate constants.
Given : k1=1×108M−1s−1(rate constant for binding)
k−1=4×104s−1(rate constant for dissociation)
k2=8×102s−1(rate constant for conversion of ES to E + P)
Step 1: Calculate Kₛ (rate constant for dissociation of enzyme-substrate complex):
The formula for Ks is given by:
Ks=k1k−1
Substitute the given values:
Ks=1×108M−1s−14×104s−1=4×10−4M=400μM
So, Kₛ = 400 μM.
Step 2: Calculate Kₘ (Michaelis constant):
The Michaelis constant Km is given by:
Km=k1k−1+k2
Substitute the given values:
Km=1×108M−1s−14×104s−1+8×102s−1=1×108M−1s−14.08×104s−1=4.08×10−4M=408μM
So, Kₘ = 408 μM.
Answer:
The correct combination of Kₛ and Kₘ is:
Kₛ = 400 μM, Kₘ = 408 μM.