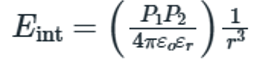Correct option is A
Ionic interactions, often referred to as ion-pairing interactions, are one of the most fundamentally important noncovalent interactions and result from electrostatic attraction between two groups of opposite charge. The strength of an ionic interaction is dictated by Coulomb’s law, in which the attractive force is a function of the distance between the charges and the dielectric constant ε of the medium.
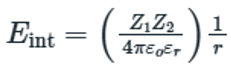
Ion (charge)–dipole interactions are fundamental attractive forces, akin to hydrogen bonds and Van der Waals interactions, that are formed between ions or charged molecules and dipolar groups with partial charges. They encompass a broad number of more specifically defined interactions, because they include charge–neutral hydrogen bonds and coordinations.
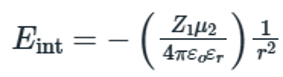
Dipole–dipole interactions are weak interactions that arise from the close association of permanent or induced dipoles. Collectively these forces are known as Van der Waals interactions.