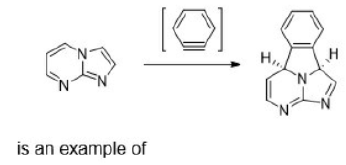
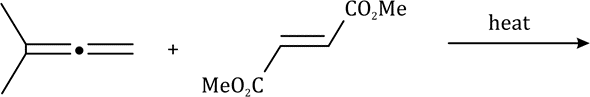Correct option is C
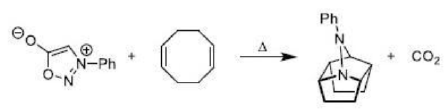
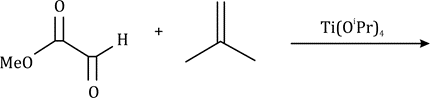
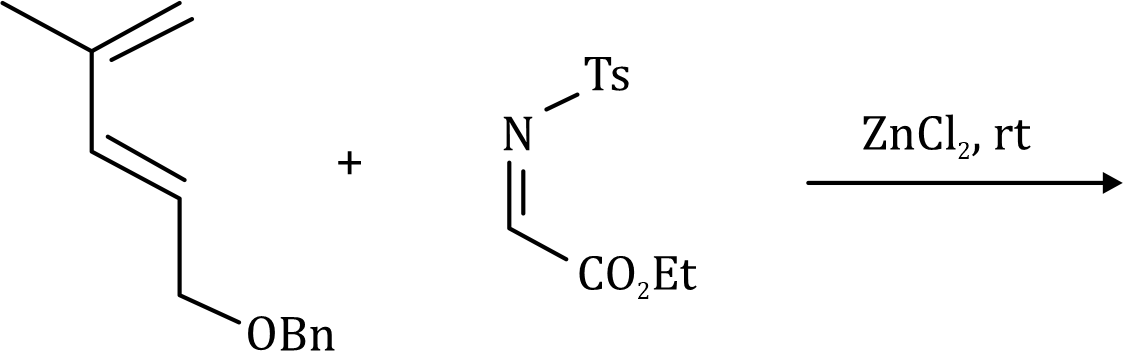
Diels–Alder reactions occur between a conjugated diene and an alkene, usually called the dienophile. It is the prototypical example of a pericyclic reaction with a concerted mechanism. Through the simultaneous construction of two new carbon–carbon bonds, the Diels–Alder reaction provides a reliable way to form six-membered rings with good control over the regio- and stereochemical outcomes.
Diels–Alder reaction is reversible and therefore under thermodynamic control, the exo product is formed.
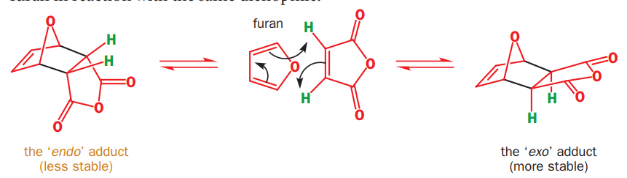
On the left-hand side of the molecules, there are two bridges across the ends of the new bonds (highlighted in black): a one-C-atom bridge and a two-C-atom bridge. There is less steric hindrance if the smaller (that is, the one-atom) bridge eclipses the anhydride ring.
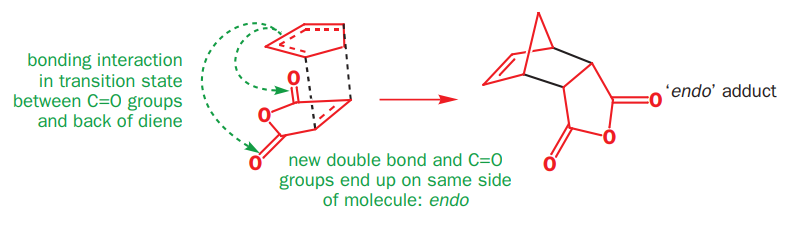
The endo product is less stable than the exo product and yet it is preferred in irreversible Diels–Alder reactions-it must be the kinetic product of the reaction. It is preferred because there is a bonding interaction between the carbonyl groups of the dienophile and the developing π bond at the back of the diene. (The black bonds are the new σ bonds between the two reagents.)
Information Booster
Thermodynamic reaction control or kinetic reaction control in a chemical reaction can decide the composition in a reaction product mixture when competing pathways lead to different products and the reaction conditions influence the selectivity or stereoselectivity. The distinction is relevant when product A forms faster than product B because the activation energy for product A is lower than that for product B, yet product B is more stable. In such a case A is the kinetic product and is favored under kinetic control and B is the thermodynamic product and is favored under thermodynamic control.
The Diels-Alder reaction of cyclopentadiene with furan can produce two isomeric products. At room temperature, kinetic reaction control prevails and the less stable endo isomer is the main reaction product. At 81 °C and after long reaction times, the chemical equilibrium can assert itself and the thermodynamically more stable exo isomer is formed. The exo product is more stable by virtue of a lower degree of steric congestion, while the endo product is favored by orbital overlap in the transition state.