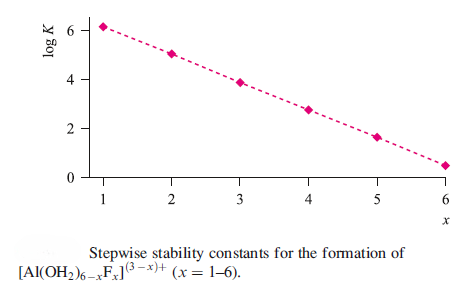
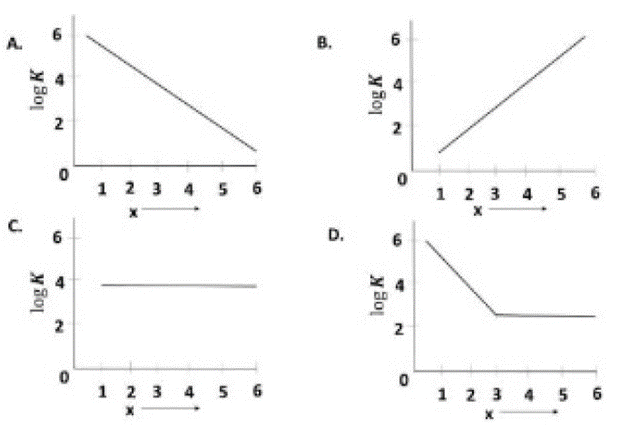
Correct option is A
Metal ions in aqueous solution are hydrated. The aqua species may be denoted as Mz+ (aq) where this often represents the hexaaqua ion [M(OH2)6]z+ . Now consider the addition of a neutral ligand L to the solution, and the formation of a series of complexes.
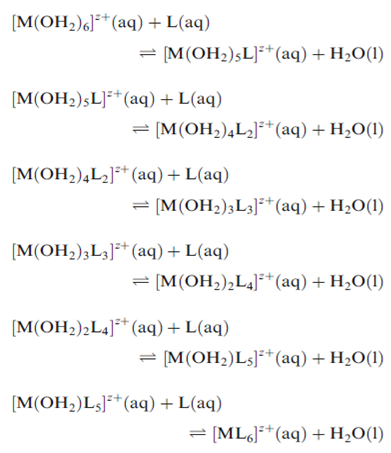

Alternatively, we may consider the overall formation of [ML6]z+. In order to distinguish stepwise and overall stability constants, the symbol β is generally used for the latter.