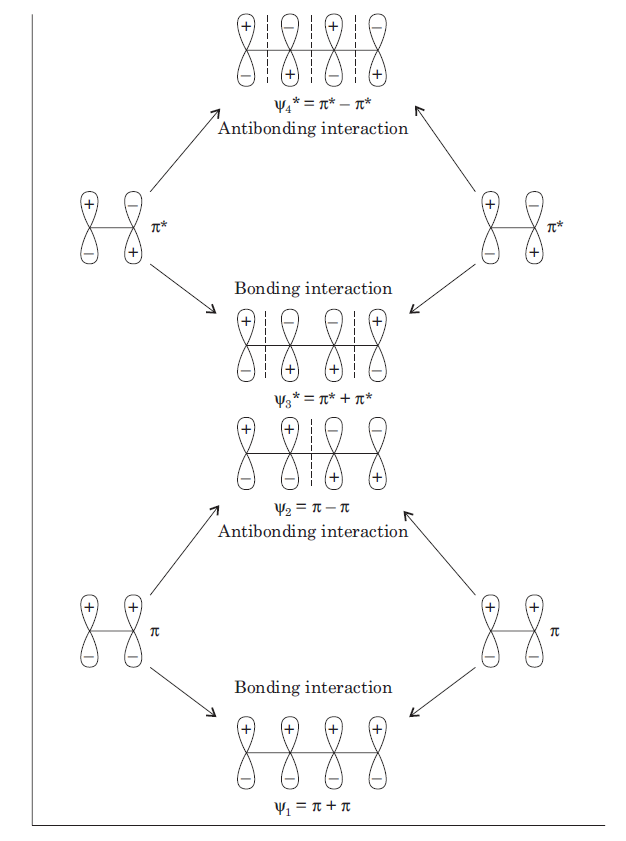
Correct option is C
In 1, 3-butadiene, we have a system of four p orbitals on four adjacent carbons. These four p orbitals will overlap to produce four π molecular orbitals. We can get four new MOs in a number of equivalent ways. One of the ways to obtain four new molecular orbitals is by linear combination of two molecular orbitals of ethylene. Linear combination of orbitals is also known as perturbation theory or perturbation molecular orbital (PMO) theory. Linear combination always takes place between two orbitals (two atomic orbitals, two molecular orbitals or one atomic and one molecular orbitals) having minimum energy difference. This means that we need to look only at the results of the π ± π and π* ± π* interactions and do not have to consider π ± π*.