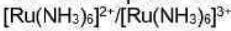Correct option is A
Covalency refers to the number of chemical bonds that an atom can form with other atoms in a molecule. It is a measure of an atom’s ability to share its electrons with other atoms to achieve stability. For many molecules, the sharing of electrons allows each atom to attain the equivalent of a full valence shell, corresponding to a stable electronic configuration.
Iodine (I) is a halogen and has seven valence electrons. Fluorine (F), also a halogen, has seven valence electrons as well. Iodine can form a maximum of seven bonds (one with each fluorine atom). Each bond corresponds to one of the valence electrons from iodine being shared with a fluorine atom.
Sulfur (S) has six electrons in its valence shell. Fluorine has seven electrons in its outermost shell. In SF6, there are six sigma bonds present between sulfur and six fluorine atoms.
Phosphorus (P) forms five single covalent bonds with five fluorine atoms. Each fluorine atom contributes one electron, and phosphorus uses its five valence electrons to form these bonds.
Silicon (Si) has four electrons in its valence shell. Fluorine has seven electrons in its outermost shell. In SiF4, there are four sigma bonds present between the silicon and four fluorine atoms.