Correct option is D
The heat of formation is the change in energy that occurs when one mole of a compound is formed from its constituent elements in their standard states usually at 25°C and 1 atm pressure. The heat of formation directly influences the stability of a compound. Compounds with lower heat of formation are more stable, as they have lower potential energy and are thermodynamically favorable. The heat of formation of a compound is the enthalpy change that occurs when one mole of the compound is formed from its elements in their standard states. It is often denoted as ΔHf. The heat of formation is a measure of the energy difference between the products and the reactants in a chemical reaction. The relationship between the heat of formation and the stability of compounds is generally straightforward: more negative values of
heat of formation indicate greater stability.
If the heat of formation is highly negative, it means that the compound is more stable because less energy is required to form it from its elements. This usually implies that the compound is thermodynamically favorable and less likely to decompose into its constituent elements. For example, noble gases have very high negative heat of formation values because they are highly stable in their elemental forms.
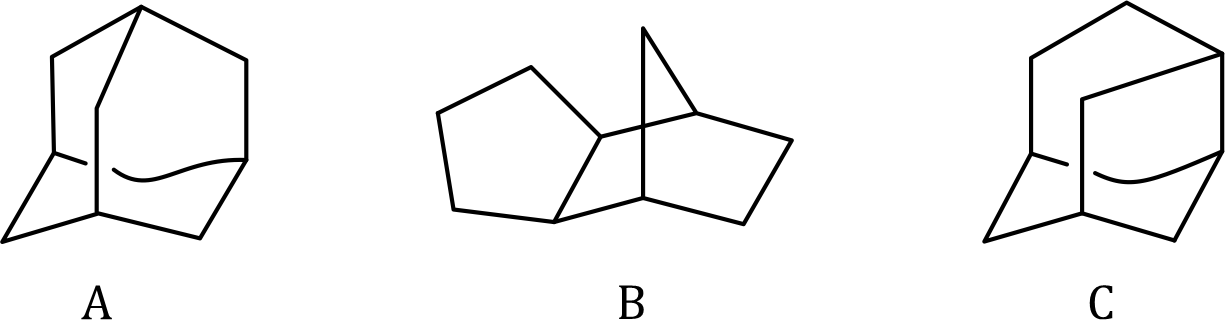
Structure A (Adamantane) is the most symmetric structure and has minimum strain. Thus, it will have the highest stability and highest heat of formation (Negative value). A has the highest amount of heat of formation amongst A, B, and C.
In comparison between structures B and C, C is more stable than B because a norbornane ring is present in B which is more strained than C. Thus, compound B will have the least stability and least heat of formation (Negative value). The heat of the formation of B and C is C> B.