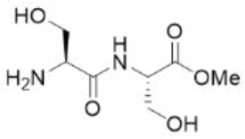
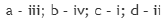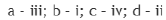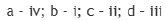Correct option is D
The bond-dissociation energy is one measure of the strength of a chemical bond A−B. It can be defined as the standard enthalpy change when A−B is cleaved by homolysis to give fragments A and B, which are usually radical species. The enthalpy change is temperature-dependent, and the bond-dissociation energy is often defined to be the enthalpy change of the homolysis at 0 K (absolute zero), although the enthalpy change at 298 K (standard conditions) is also a frequently encountered parameter.
Electronegativity and hybridization affect the bond dissociation energy. Bond strength is directly proportional to s character. Higher the bond strength, higher will be the bond dissociation energy.
Electronegativity, symbolized as χ, is the tendency for an atom of a given chemical element to attract shared electrons (or electron density) when forming a chemical bond. An atom's electronegativity is affected by both its atomic number and the distance at which its valence electrons reside from the charged nucleus. The higher the associated electronegativity, the more an atom or a substituent group attracts electrons. Electronegativity serves as a simple way to quantitatively estimate the bond energy, and the sign and magnitude of a bond's chemical polarity, which characterizes a bond along the continuous scale from covalent to ionic bonding.
The electronegativity of an atom changes depending on the hybridization of the orbital employed in bonding. Electrons in s orbitals are held more tightly than electrons in p orbitals. Hence, a bond to an atom that employs an spx hybrid orbital for bonding will be more heavily polarized to that atom when the hybrid orbital has more s character. That is, when electronegativities are compared for different hybridization schemes of a given element, the order χ(sp3) < χ(sp2) < χ(sp) holds.
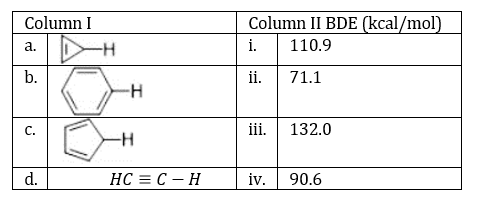
The carbon atom in the C-H bond in the structures a, b, c and d are sp3,sp2,sp3 and sp hybridized respectively.
sp hybridization: s and p each contribute 50 %. The bond angle is 180 degrees.
sp2hybridization: In this type of hybridisation, s contributes 33.33%, while p contributes 66.66%.The bond angle is 120 degrees.
sp3hybridization: In this hybridisation, s contributes just 25%, and p contributes around 75%. The bond angle is 109.5 degrees.
Angle strain arises when the C-C-C bonds of the ring depart (because of geometric necessity) from the ideal tetrahedral angle preferred for sp3 carbon. Angle strain in cyclopropane arises from the circumstance that the C-C-C angle is forced to be 60 degrees (that of an equilateral triangle), about 49 degrees less than the ideal angle for the strongest C-C bond.
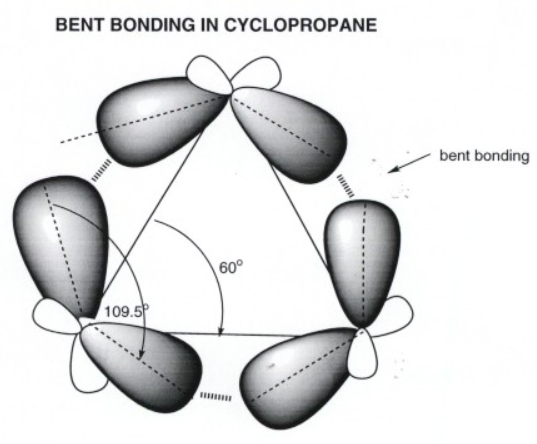
The s character reduces in the C-C bonds and is shifted to the C-H bond. Hence, the carbon atom in the C-H bond in structure a has more s character than the carbon atom in the C-H bond in structure c.