Correct option is C
The notation for a full term symbol is:
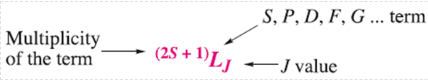
The energy and the orbital angular momentum of a multielectron species are determined by a quantum number, L. Energy states for which L=0, 1, 2, 3, 4... are known as S, P, D, F, G... terms, respectively.
For any system containing more than one electron, the energy of an electron with principal quantum number n depends on the value of l, and this also determines the orbital angular momentum which is given by the equation:
Planck constant is expressed in SI units, it has the exact value h=6.626×10-34JHz-1
The spin quantum number, s, determines the magnitude of the spin angular momentum of an electron and has a value of 1/2. For a 1-electron species, ms is the magnetic spin angular momentum and has a value of +1/2 or -1/2. The spin angular momentum for a multielectron species is given by the following equation, where S is the total spin quantum number. The quantum number MS is obtained by algebraic summation of the ms values for individual electrons:
For each value of S, there are (2S+1) values of MS.

The interaction between the total angular orbital momentum, L, and the total spin angular momentum, S is defined by the total angular momentum quantum number, J.
The following equation gives the relationship for the total angular momentum for a multi-electron species.
The quantum number J takes values (L+S), (L+S-1).....L-S, and these values fall into the series 0,1,2....or 1/2,3/2,5/2. The method of obtaining J from L and S is based on LS (or Russell-Saunders) coupling, that is spin-orbit coupling. The value of J for the ground state is given by (L-S) for a sub-shell that is less than half-filled, and by (L+S) for a sub-shell that is more than half-filled.
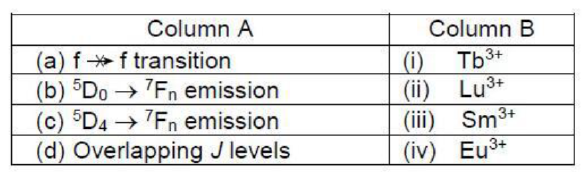
Lu3+ion ([Xe]4f14) has completely filled orbitals and the f-f transition is spin forbidden (ΔS is not equal to zero). Hence, there is no transition.
The Eu3+ ion has a 4f6 electron configuration and S = 3. The term with the lowest energy is 7F with L = 3. Strong spin-orbit coupling results in a J = 0 ground state. As the Eu3+ ion has a less than half-filled electronic configuration, the value of J will be |3-3| = 0. Thus, an energy state with the term symbol 5D0, is possible for the Eu3+ ion.
5D0 → 7Fn, emission is observed in the Tb3+ ion. The Tb3+ ion has a 4f8 electronic configuration and S = 3. The term with the lowest energy is 7F with L = 3. Strong spin-orbit coupling results in a J = 6 ground state. As the Tb3+ ion has a more than half-filled electronic configuration, one possible value of J can be = L+S-2 =3+3-2 = 4. Thus, an energy state with the term symbol 5D4 is possible for the Tb3+ ion.
Sm3+ ion has a 4f5 electronic configuration. At room temperature, the first, second and third excited state are found to be populated. Mixing of these states with higher J values causes the larger deviation in observed magnetic moment because of overlapping J levels.