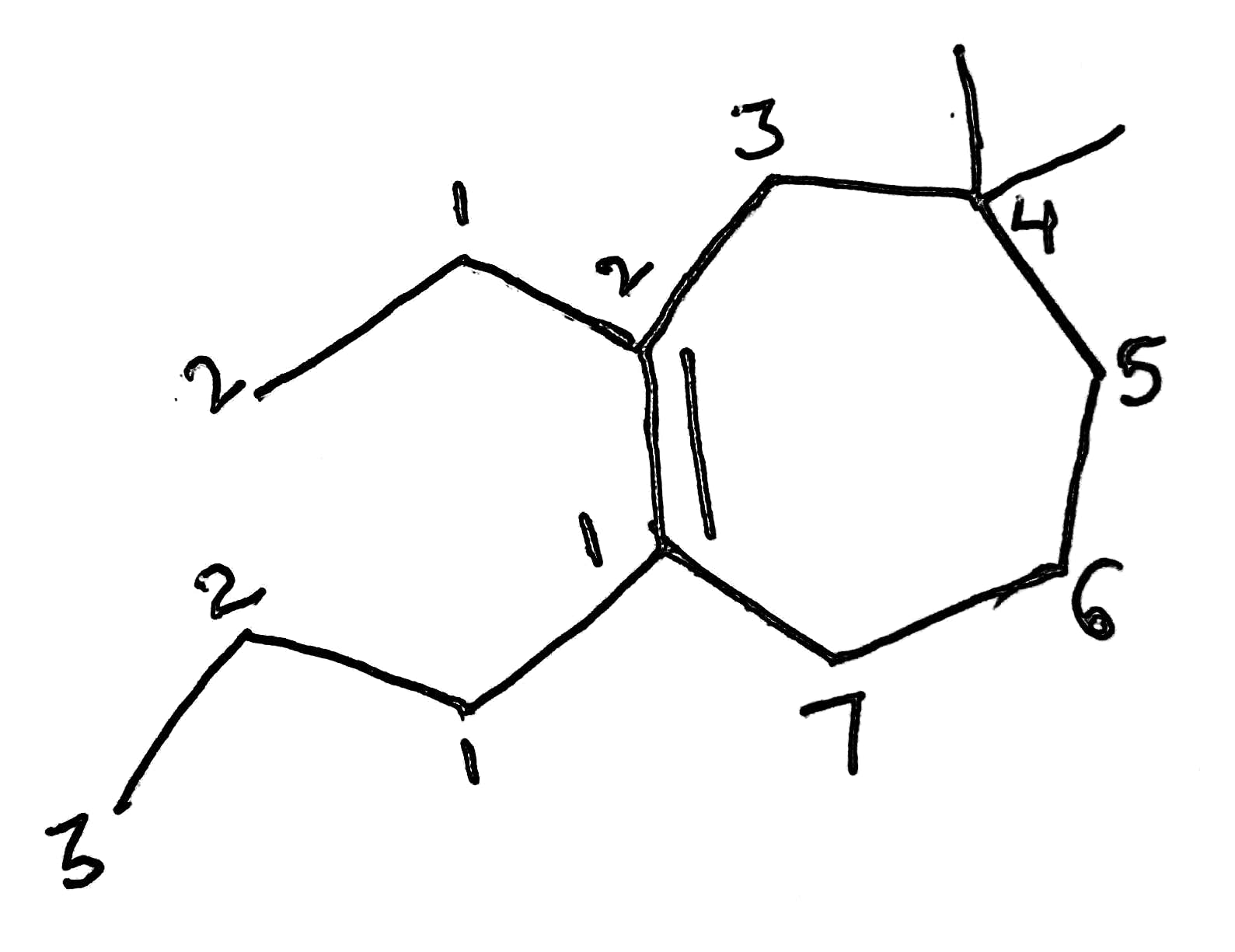
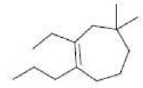
Correct option is B
In chemical nomenclature, the IUPAC nomenclature of organic chemistry is a method of naming organic chemical compounds as recommended by the International Union of Pure and Applied Chemistry (IUPAC).
Basic Principles
The steps for naming an organic compound are:
Identification of the ring or chain with the maximum number of senior groups.
Identification of the parent compound. Rings are senior to chains if composed of the same elements.
For cyclic systems and chains:
It should have the maximum number of multiple, then double bonds.
Identification of the side-chains. Side chains are the carbon chains that are not in the parent chain, but are branched off from it. The multiplier prefixes di-, tri-, etc. are not taken into consideration for grouping alphabetically. For example, ethyl comes before dihydroxy or dimethyl, as the "e" in "ethyl" precedes the "h" in "dihydroxy" and the "m" in "dimethyl" alphabetically. The "di" is not considered in either case.
Identification of double/triple bonds.
Numbering of the various substituents and bonds with their locants. If there is more than one of the same type of substituent/double bond, a prefix is added showing how many there are (di-2, tri-3, tetra-4, then as for the number of carbons below with 'a' added at the end). The numbers for that type of side chain will be grouped in ascending order and written before the name of the side-chain. If there are two side-chains with the same alpha carbon, the number will be written twice. Example: 2,2,3-trimethyl- . If there are both double bonds and triple bonds, "en" (double bond) is written before "yne" (triple bond).
Adding of punctuation:
Commas are put between numbers (2 5 5 becomes 2,5,5)
Hyphens are put between a number and a letter (2 5 5 trimethylheptane becomes 2,5,5-trimethylheptane)
Successive words are merged into one word (trimethyl heptane becomes trimethylheptane)
Note: IUPAC uses one-word names throughout. This is why all parts are connected.
In the given compound, the principal functional group is an alkene.
The cyclic ring consists of longest continuous chain of carbon atoms and is thus the parent chain.
Alkenes are named for their parent alkane chain with the suffix "-ene" and a numerical root indicating the position of the carbon with the lower number for each double bond in the chain.
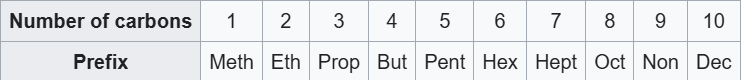
As the chain consists of seven carbon atoms it is named as heptene.
Out of the two groups ethyl and propyl, the atomic mass of the propyl group is higher than ethyl. So propyl group gets higher priority over the ethyl.