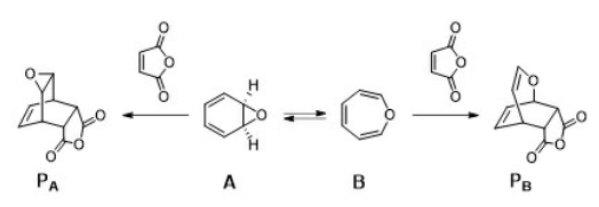
Correct option is C
In organic chemistry, the Diels–Alder reaction is a chemical reaction between a conjugated diene and a substituted alkene, commonly termed the dienophile, to form a substituted cyclohexene derivative. It is the prototypical example of a pericyclic reaction with a concerted mechanism.
A Diels-Alder system that is more easily studied is the dimerization of 1,3-cyclopentadiene shown below.

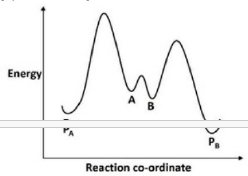
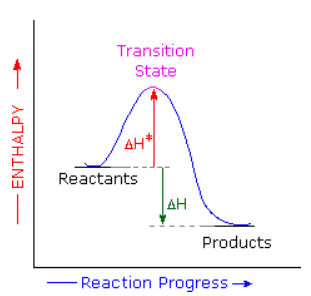
The availability of activation parameters for this reaction makes it possible to explore further details of the reaction path. The stereospecificity of the Diels-Alder reaction implies a single step transformation, and the exothermic character of the reaction requires an enthalpy profile similar to that shown below. The Hammond Postulate, which uses ΔHº as an indicator, suggests that the transition state would resemble the reactants more than the products. However, the similar magnitude and sign of ΔSº and ΔS‡ requires the transition state to have a structure very similar to the dimer product.
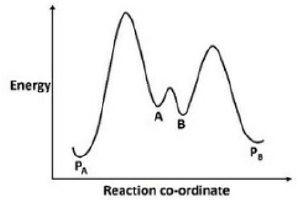
In the given question, compound A is less stable and more reactive. Compound A has a smaller ring and thus acts as a more rigid diene. Compound B is a seven-membered ring. Bigger rings are more flexible. Compound B will have less reactivity towards the dienophile.
Activation energy will be lower for the formation of PA.