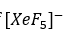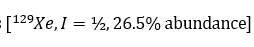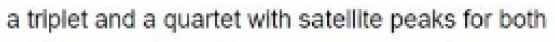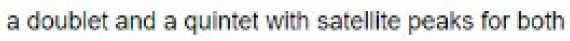Correct option is D
Nuclear magnetic resonance (NMR) spectroscopy is a resonance technique involving absorption of radiofrequency energy. The magnetic environment of a nucleus affects its resonance frequency and allows structural information to be deduced.
Structural assignment is often helped by the observation of the spin–spin coupling, which gives rise to multiplets in the spectrum due to interactions between nuclear spins. Spin–spin coupling arises when the orientation of the spin of a nearby nucleus affects the energy of another nucleus and causes small changes in the location of the latter’s resonance. A multiplet of 2I+1 lines is obtained when a spin-1/2 nucleus (or a set of symmetry-related spin-1/2 nuclei) is coupled to a nucleus of spin I. The coupling of the nuclear spins of different elements is called heteronuclear coupling. Homonuclear coupling between nuclei of the same element is detectable when the nuclei are in chemically inequivalent locations.
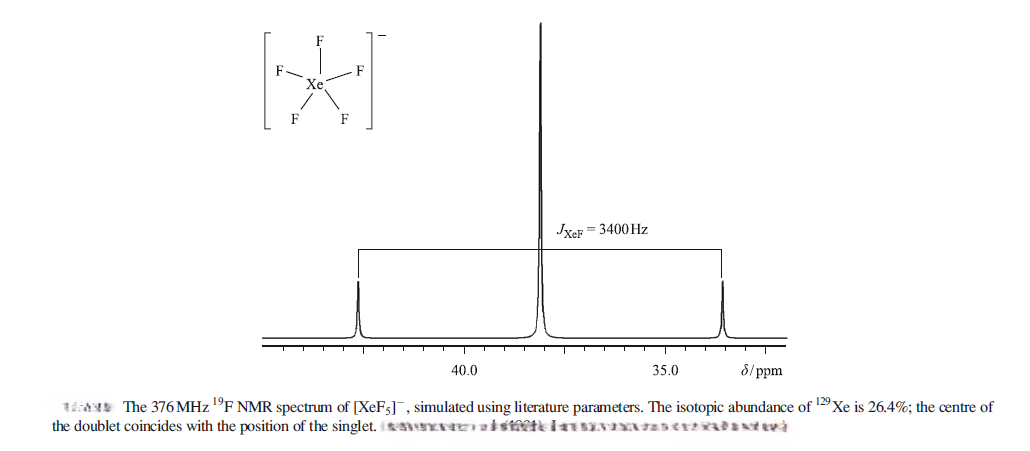
The planar [XeF5 ]- ion contains five equivalent F atoms. Both the 19F and 129Xe nuclei are NMR active: 19F, I=1/2, 100% abundance; 129Xe, I=1/2, 26.4%. The chemical equivalence of the 19F nuclei gives rise to one signal. However, 26.4% of the F centres are attached to 129Xe, while the remainder are bonded to other Xe nuclei. The spectrum can be interpreted in terms of a singlet (the central line) due to 73.6% of the 19F nuclei, plus an overlapping doublet due to the 26.4% of the 19F nuclei that couple to 129Xe. The centre of the doublet coincides with the position of the singlet because all the 19F nuclei resonate at the same frequency. The two side peaks are called satellite peaks.
Major splitting ( 129Xe, I=0, 73.6%):2×0+1=1
Minor splitting ( 129Xe, I=1/2, 26.4%):2× (1/2) +1=2