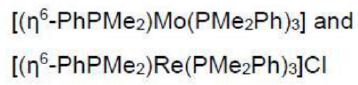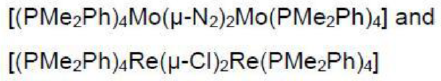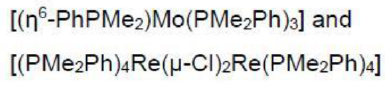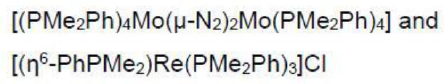Correct option is C
The 18-electron rule is a chemical rule of thumb used primarily for predicting and rationalizing formulas for stable transition metal complexes, especially organometallic compounds. The rule is based on the fact that the valence orbitals in the electron configuration of transition metals consist of five (n−1)d orbitals, one ns orbital, and three np orbitals, where n is the principal quantum number. These orbitals can collectively accommodate 18 electrons as either bonding or non-bonding electron pairs. This means that the combination of these nine atomic orbitals with ligand orbitals creates nine molecular orbitals that are either metal-ligand bonding or non-bonding. When a metal complex has 18 valence electrons, it is said to have achieved the same electron configuration as the noble gas in the period, lending stability to the complex. Transition metal complexes that deviate from the rule are often interesting or useful because they tend to be more reactive.
Neutral-ligand method: For the sake of counting electrons, each metal atom and ligand is treated as neutral. We include in the count all valence electrons of the metal atom and all the electrons donated by the ligands. If the complex is charged, we simply add or subtract the appropriate number of electrons to the total. The advantage of the neutral-ligand method is that, it is trivial to establish the electron count. The disadvantage, however, is that the method overestimates the degree of covalence and thus underestimates the charge at the metal. Moreover, it becomes confusing to assign an oxidation number to a metal, and meaningful information on some ligands is lost.The total electron count is the sum of the number of electrons on the metal atom and the number of electrons provided by the ligands.
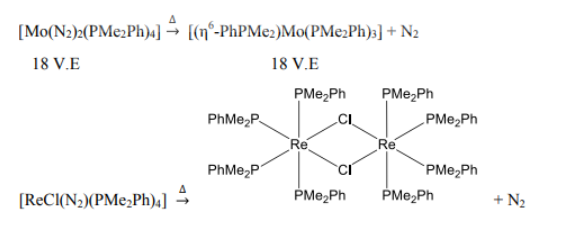
Mo is six electron donor. Dinitrogen is a two electron donor. PMe2Ph is a two electron donor. η6-PhPMe2 is a six electron donor.
Re is a seven electron donor. The Cl in the reactant is a one electron donor. In the product, each Cl ligand donates three electrons (bridging).