Correct option is C
The valence shell of a lanthanoid element contains 4f orbitals. The 4f atomic orbitals are deeply buried and 4f electrons are not available for covalent bonding. Usually for a lanthanoid metal, M, ionization beyond the M3+ion is not energetically possible. This leads to a characteristic +3 oxidation state across the whole row from La to Lu.
For the lanthanoids, spin–orbit coupling is more important than crystal field splitting, and terms differing only in J values are sufficiently different in energy to be separated in the electronic spectrum. Further, since l=3 for an f electron, ml may be 3, 2, 1, 0, -1, -2 or -3, giving rise to high values of L for some fn ions. Since S, P, D, F and G terms are also possible, many of them with different positive values of J, the number of possible transitions is large, even after taking into account the limitations imposed by selection rules. As a result, spectra of Ln3+ions often contain large numbers of absorptions. Since the 4f electrons are well shielded and not affected by the environment of the ion, bands arising from f–f transitions are sharp (rather than broad like d–d absorptions) and their positions in the spectrum are little affected by complex formation. Intensities of the absorptions are low, indicating that the probabilities of the f–f transitions are low, i.e. little d–f mixing. Absorptions due to 4f–5d transitions are broad and are affected by ligand environment.
Information Booster
The notation for a full term symbol is:
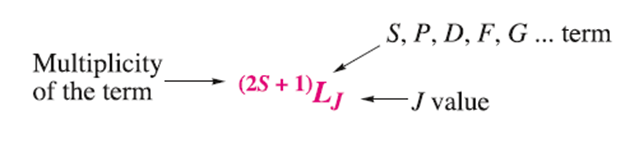
The energy and the orbital angular momentum of a multielectron species are determined by a quantum number, L. Energy states for which L=0, 1, 2, 3, 4... are known as S, P, D, F, G... terms, respectively.
For any system containing more than one electron, the energy of an electron with principal quantum number n depends on the value of l, and this also determines the orbital angular momentum which is given by the equation:

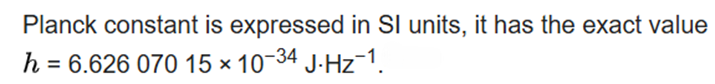
The spin quantum number, s, determines the magnitude of the spin angular momentum of an electron and has a value of 1/2. For a 1-electron species, ms is the magnetic spin angular momentum and has a value of +1/2 or -1/2. The spin angular momentum for a multielectron species is given by the following equation, where S is the total spin quantum number.
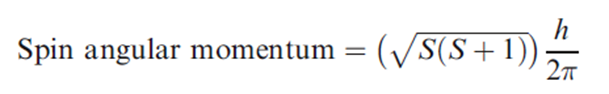
The quantum number MS is obtained by algebraic summation of the ms values for individual electrons:
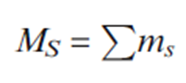
For each value of S, there are (2S+1) values of MS

The interaction between the total angular orbital momentum, L, and the total spin angular momentum, S is defined by the total angular momentum quantum number, J.
The following equation gives the relationship for the total angular momentum for a multi-electron species.

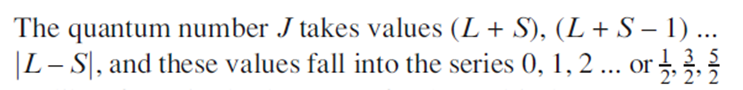

The value of J for the ground state is given by (L-S) for a sub-shell that is less than half-filled, and by (L+S) for a sub-shell that is more than half-filled.